A group of Turkish university researchers has assessed the viability of using 3D bioprinting technology to enhance success rates in spinal surgery.
By combining CT scanning and 3D bioprinting technologies, the research team was able to rapidly convert anatomical images into a series of guides and 3D models. The novel components could allow for new applications in spinal surgery, including preoperative planning, surgical simulation, and potentially implantable devices.
Additive manufacturing in spinal surgery
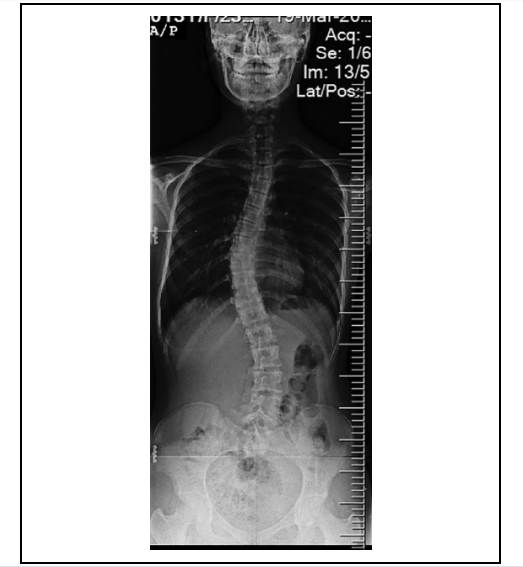
Developing a sound knowledge of human anatomy and the ability to identify landmarks during surgery, can involve a steep learning curve, but these are important requirements for proper surgical intervention. According to the researchers, traditional intraoperative techniques such as CT scanning and fluoroscopy-based methods can lead to prolonged surgery duration, radiation exposure, and high costs.
The research team suggests that utilizing haptic sensation rather than 2D images to assess patients, could provide spinal surgeons with more realistic anatomical demonstrations of patients’ deformities. Using this more detailed approach could lead to reductions in surgical duration, blood loss, and fluoroscopy time, as well as costs, by eliminating intraoperative navigation usage in complex spinal procedures.
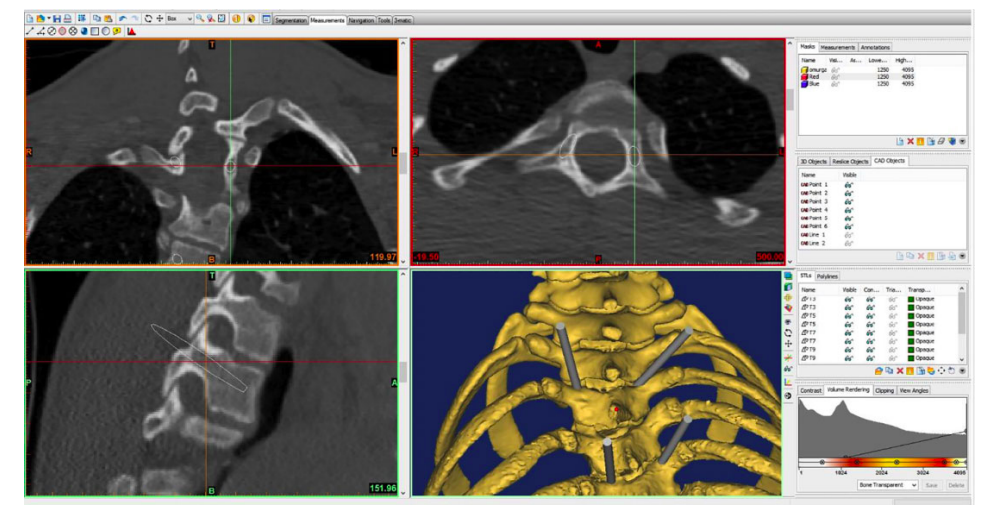
Operations for treating spinal deformities often require the production of ready to use implants for anterior column reconstruction, which brings the risk of implant dislodgement following an inaccurate fit. 3D printing these parts, gives them an even load distribution and superior osseointegration, allowing implants to be tailor-made for patients, and reducing the risk of complications. The ‘DP models’ produced using this process, were a more accurate representation of a patient’s anatomy and showed less common anatomical structures.
3D printing’s core layer-by-layer concept enabled the researchers to produce detailed anatomical models, which would not be possible using other methods, even sophisticated milling machines. Moreover, the researchers found that fewer industrial machines were required to begin manufacturing, and that little training was needed to operate the systems. Combining additive manufacturing with CT scanning technology, therefore, allowed for the production of complex models with both external and internal contour geometries, that could closely resemble those of a patient’s anatomy.
Additive advances in spinal surgery
Pedicle screw insertion is a common procedure in spinal surgery, but it’s fraught with potentially devastating and life-threatening complicated injuries of the inferior vena cava, descending aorta, spinal cord, and lung. Korean researchers found in 2018, that 3D printing could be used to avert the risks of such complications. In their study, residents with no prior knowledge of the insertion procedures were able to implement the procedure on a 3D printed model. Although the imitations couldn’t fully mimic the texture of real human tissues, the design was found to provide a better representation of anatomy than standard imaging methods and had the potential to improve doctor-patient interactions.
Using 3D printing could also help to reduce complications in screw insertion surgery, by helping surgeons to determine the pedicle entry points, and accurate trajectories for insertion through 3D printed guides. Based on preoperative CT scans or MRI imaging of the patient, the guide layout consists of a segment that matches the posterior vertebral bone structures and an outlined drilling trajectory for screw instrumentation. In an operation scenario, the sterilized component is put onto the surface of the target vertebra, to correct the pedicle screw direction.
During a pilot study, the procedure was carried out using 3D guides on eleven patients. The components enabled preoperative planning, facilitated pedicle screw insertion intraoperatively, and the method was found to be economical and accessible. Similarly, a study carried out in 2019 used 3D printed guides on a series of 15 severe congenital scoliosis patients. These researchers surmised that screw insertion was more accurate than the steadiest freehand technique, and recommended the guides to those surgeons that do not have access to CT scanners or O-arm navigation. Moreover, five abnormal or variant pedicles were encountered. The utilization of guide templates helped surgeons to pick the correct screws, and choose the most ideal screw trajectory for every pedicle, to effectively deal with the variations.
The Turkish researchers concluded that using 3D printed guides leads to increased patient safety, allows surgeons to perform more complicated operations, and enables the creation of customized implants for bone defects. Future applications could be in preoperative planning, surgical simulations, and intraoperative guidance. While the research team acknowledged concerns about debris produced by polymer-based templates, titanium patient-specific templates were found to resolve this issue, albeit at a higher cost. The researchers anticipate further studies to continue developing the technology and find different aspects of spinal surgery on which to use it.
Additive applications in surgery
Various other 3D printing companies have innovated their own solutions to producing spinal implants, with many of them gaining the U.S Food and Drug Administration (FDA) approval.
Texan medical device company Osseus Fusion Systems received FDA approval for its Aries range of 3D printed spinal implants in August 2018. Produced using titanium material, the Aries lumbar interbody fusion devices are designed to help surgeons ease back pain and shorten spinal recovery time after surgery.
Centinel Spine, a Pennsylvania-based spinal device development company, received clearance from the FDA in June 2018, to market its 3D printed implantation devices. The company’s FLX Platform consists of all-titanium devices that work to stabilize vertebrae in the spine, and increase the healing process for patients after spinal surgeries.
Medical firm Emerging Implant Technologies GmbH (EIT) gained FDA approval in January 2018 to produce the “first-ever” 3D printed spine implant for treating multiple injuries. Using selective laser melting (SLM) 3D printing allowed EIT to make implants with a core that mimics the cancellous (“spongy”) and cortical (hard) structure of bone.
The researchers’ findings are detailed in their paper titled “3D printing and spine surgery,” published on May 29th, 2020 in the Journal of Orthopaedic Surgery. The research was co-authored by Alpaslan Senkoylu, Ismail Daldal, and Mehmet Cetinkaya.
You can now nominate for the 2020 3D Printing Industry Awards. Cast your vote to help decide this year’s winners.
To stay up to date with the latest 3D printing news, don’t forget to subscribe to the 3D Printing Industry newsletter or follow us on Twitter or liking our page on Facebook.
Looking for a job in the additive manufacturing industry? Visit 3D Printing Jobs for a selection of roles in the industry.
Featured image shows the silhouette of a woman experiencing pain in her spine. Photo via the Royal Society of Physiotherapy.



