Researchers from Carleton University and the University of Northern British Columbia have demonstrated the value of titania quantum dots for resin 3D printing.
Photopolymerization is a key step in stereolithography (SLA) 3D printing. However, conventional photoinitiators used in this process are toxic, expensive, and can’t be easily adjusted to different light wavelengths. Significantly, organic photoinitiators that are cured with UV degrade under sunlight, negatively impacting the quality of the 3D printed part.
In a recent study published in the journal Industrial Chemistry & Materials, the Canada-based researchers have overcome these common issues.
The team found that titania quantum dots, which are non-toxic and inexpensive, can initiate photopolymerization in epoxy resin when exposed to UVC light, which is not in natural sunlight. The quantum dots also remain stable when subjected to UVA light, which is found in sunlight, ensuring the resin-3D printed parts don’t degrade over time.
This study was co-authored by Keroles Riad, and builds on his previous research into the photostability of materials used in SLA 3D printing.
Speaking to 3D Printing Industry in 2016, the then Carleton University PhD student explained that “The whole stability problem with SLA resin is because the same UV you print with is also present in the solar spectrum. Initiating the [photocuring] reaction with wavelengths outside the solar spectrum would solve this.”
According to the researchers, their new approach “allows precise control over the wavelengths of light under which photopolymerization can and cannot occur,” improving the outcomes of SLA 3D printing.

Sunlight instability in SLA 3D printing
During photopolymerization, light is directed at a liquid resin, triggering a chemical reaction that hardens or “cures” the polymer through crosslinking (or bonding). This is repeated throughout the SLA 3D printing process, solidifying resin layer by layer until the final part has been fabricated.
Conventional epoxy resins used in SLA 3D printing are unstable when exposed to sunlight. This is because the absorption wavelength range of commercial photoinitiators, which trigger the polymerization process, overlaps with the solar spectrum on Earth.
These organic photoinitiators absorb UVA and UVB light, which make up around 6% of sunlight. As such, the 3D printed parts continue to polymerize when exposed to natural light. This process releases harmful chemicals and can cause the parts to become brittle and break in a matter of weeks.
The toxicity of conventional photoinitiators poses significant challenges for the photopolymer market. Last year, the European Chemicals Agency announced its intention to identify diphenyl(2,4,6-trimethylbenzoyl)phosphine oxide (TPO), a popular photoinitiator, as a Substance of Very High Concern.
UV absorbers, light stabilizers and inorganic surface coatings can be used to reduce the instability of these SLA-3D printed parts. However, the researchers liken these options to sunscreen, as they only delay the degradation and must be regularly reapplied.
The team points to the Earth’s ozone layer as providing the answer to this problem, as it blocks light of wavelengths lower than 300 nm (UVC light). Therefore, they believe photocatalysts that are sensitive to light with sub-300 nm wavelengths hold the key to producing more stable parts with SLA 3D printing.
Improving SLA 3D printing with quantum dots
Quantum dots are tiny nanoparticles that possess semiconducting properties. During SLA 3D printing, they absorb light and become excited, transferring their energy to the surrounding resin. This energy triggers polymerization.
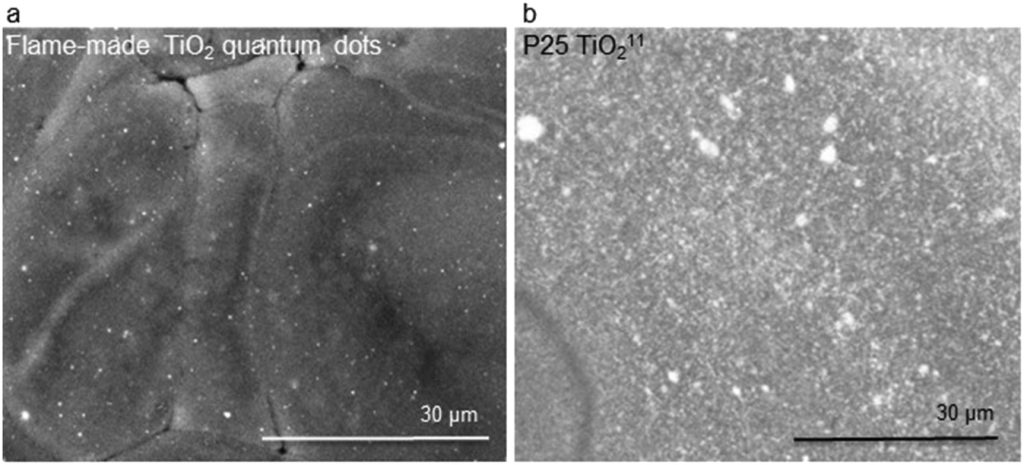
In the study, the researchers synthesized their titania quantum dots using the flame spray pyrolysis (FSP) process. Here, a flame is used to heat a liquid solution. The resulting reaction causes the liquid ingredients to form nanoparticles.
The team added silica to this process to create their metal oxide quantum dots. This hinders the growth of the metal oxide crystals, resulting in crystal sizes small enough to show quantum effects.
The nanoparticles were created with 10% TiO2 and 90% silica. The significant TiO2 crystalline fraction of the flame-made titania nanoparticles made it 2-3 times more efficient in catalyzing epoxy photopolymerization than P25 titania, “the gold standard for photocatalysis.”
According to the researchers, their quantum dots are affordable to produce and do not share the toxicity of conventional photoinitiators. Indeed, titania is a readily available material, with over 2 million tons produced yearly. Thanks to its lack of toxicity, it is a common ingredient in white paint and coatings of medical pills
Notably, the wavelength of light these particles are sensitive to can be determined by changing their size. As such, the researchers’ quantum dots were designed to be sensitive to UVC light, and unaffected by the wavelength of natural sunlight.
Are quantum dots better than organic photoinitiators?
In the study, epoxy films were 3D printed using conventional organic photoinitiators and the researchers’ quantum dots. Nuclear magnetic resonance spectroscopy (NMR) and Fourier Transform Infrared Spectroscopy (FTIR) imaging were used to demonstrate that photopolymerization was initiated under different light sources.
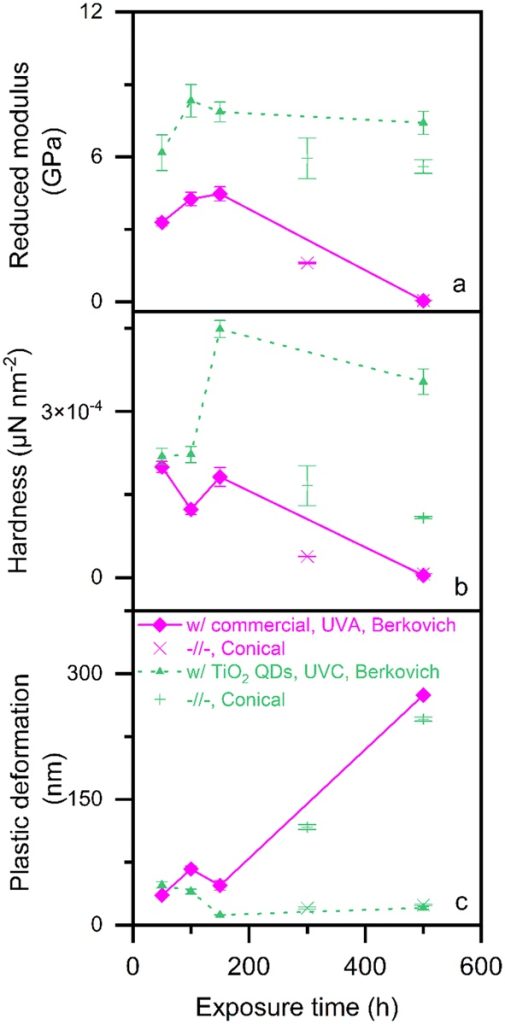
Nanoindentation was then used to monitor the mechanical properties of the 3D printed epoxy films when exposed to UVA light. These properties include reduced modulus, hardness, and plastic deformations.
The 3D printed films photopolymerized with quantum dots exhibited significantly better properties than those with a commercial organic photoinitiator. After 150 hours of sunlight exposure, the 3D printed with quantum dot resin parts showed double the reduced modulus and hardness, and 75% less plastic deformation.
After 300 hours of UVA exposure, the epoxy films photopolymerized with the commercial organic photoinitiator drastically deteriorated, with reduced modulus and hardness dropping to near zero. Alternatively, those photopolymerized with titania quantum dots remained stable for 500 hours of UVA exposure.
As such, the research team concluded that “UVA light, and in turn, sunlight, has no effect on the mechanical properties of the epoxy films photopolymerized with titania quantum dots.”
3D printing quantum dots
The Carleton University and University of Northern British Columbia researchers are not alone in recognising the value of quantum dots is 3D printing.
Back in 2018, a team from the University of Miami created a luminescent 3D printing material that featured quantum dots. Designed to defend against damaging rays of UV light, the SLA 3D printing resin was made by mixing glowing, orange carbon dots (O-CDs) with a superabsorbent polymer.
To assess the utility of this 3D printing ink, the researchers 3D printed small models of the Statue of Liberty using a Formlabs desktop SLA system. Here, the team discovered that O-CD inks can be 3D printed without affecting their photoluminescence (PL). As such, they noted that the material offers potential for UV light converters, sunglasses and probes for environmental, biological or medical purposes.
Elsewhere, researchers from the Korea Electrotechnology Research Institute have used quantum dots to 3D print high-density pixel arrays at the nanoscale. The team embedded and solidified liquid inks in polymer nanowires to fabricate tiny freestanding pillars capable of emitting light of different colors.
Quantum dots that had peak emission wavelengths of 650, 540, and 480 nm (corresponding to red, green, and blue) were added to solvated polystyrene polymers. The resulting nano ink was 3D printed via direct writing onto a flat quartz plate substrate to create the RGB display. The researchers reportedly chose quantum dots due to their high-quantum efficiencies and relatively long-term stability.
Want to help select the winners of the 2024 3D Printing Industry Awards? Join the Expert Committee today.
What does the future of 3D printing hold?
What near-term 3D printing trends have been highlighted by industry experts?
Subscribe to the 3D Printing Industry newsletter to keep up to date with the latest 3D printing news.
You can also follow us on Twitter, like our Facebook page, and subscribe to the 3D Printing Industry Youtube channel to access more exclusive content.
Featured image shows a TEM image of the flame-made silica-embedded TiO2 quantum dots used in this study. Image via Industrial Chemistry & Materials.


