Researchers at the University of California in Los Angeles (UCLA) have developed a viable bioink for 3D printed pills.
As seen at the 3D Medical Expo in Maastricht, 3D printed pharmaceuticals are a relatively new venture but believed to have great potential for innovating an industry that has stayed the same since its conception.

Personalized healthcare
The apparent disadvantage of current medication is that it doesn’t take a person’s specific genetic make-up or blood content into account. As everyone’s genome is unique, the way the body absorbs a drug also varies from person to person.
Referencing figures published by Nature in 2015, the paper Photocurable Bioink for the Inkjet 3D Pharming of Hydrophilic Drugs states that “the top 10 highest grossing medications only have a positive therapeutic effect in 4–25 percent of the patients that take them”.
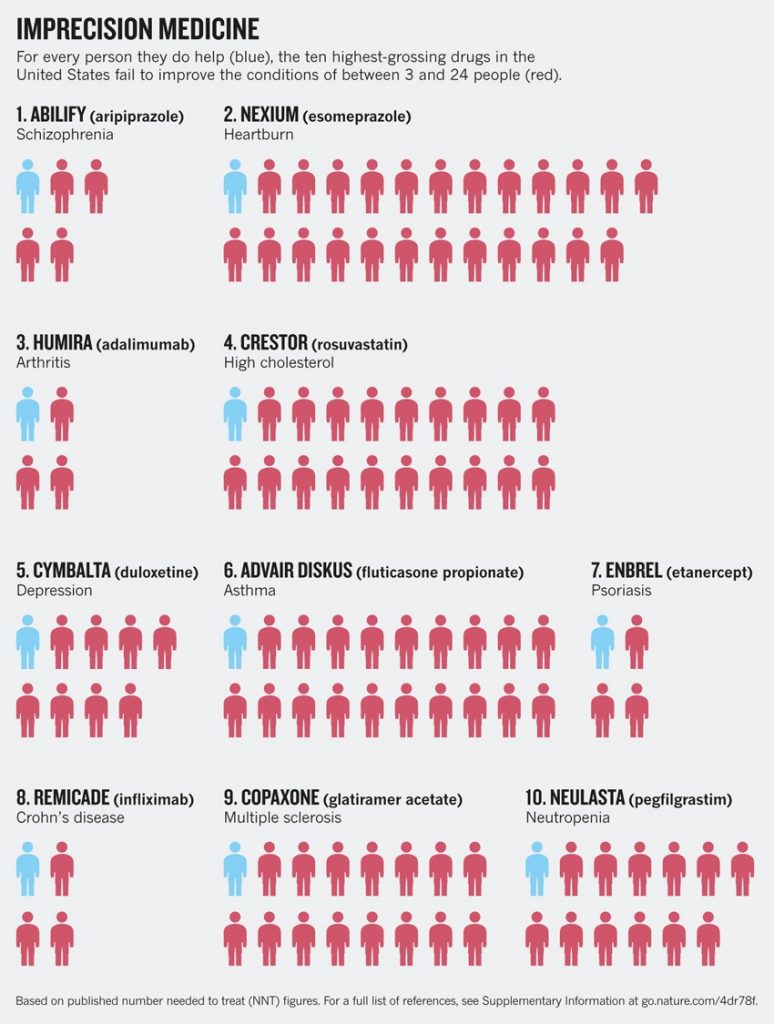
Nicholas J. Schork
3D printed pill production
In this study, Giovanny Acosta-Vélez, Chase Linsley, Madison Craig and Benjamin Wu favour the inkjet technique over other 3D printing technologies for its speed and ability to print at room temperature. The temperate environment ensures that active pharmaceutical ingredients (APIs) aren’t damaged in the process, and speed is of course preferable for mass production.
The 3D printable bioink from UCLA is made from hyaluronic acid – a key ingredient in skin, connective tissues and the nervous system. A photoinitator is added to the acid so that it solidifies when in contact with light. This mixture is used to fill preformed tablets displaying the properties of an atypical oral tablet.
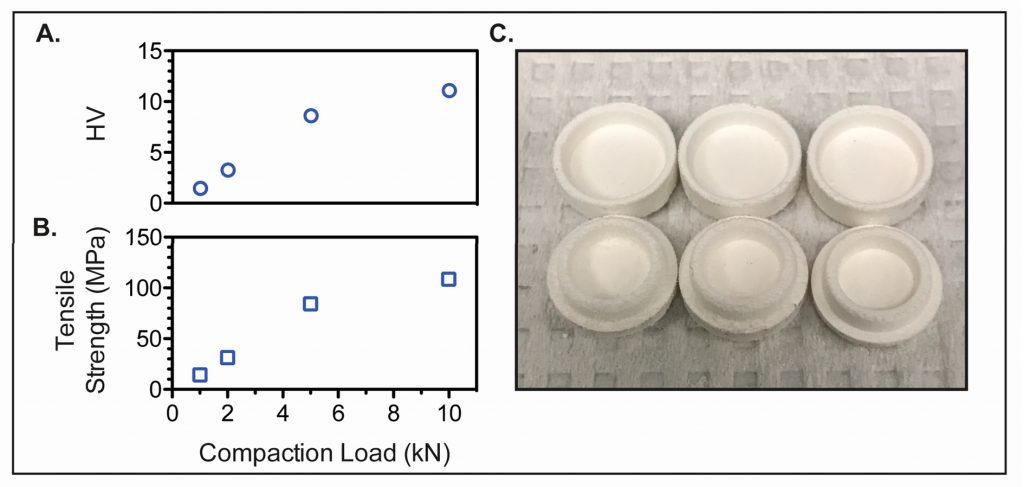
The hyaluronic acid is then laced with Ropinirole Hcl – a stimulant commonly used for the therapeutic treatment of muscle problems associated with Parkinson’s disease. The Ropinirole Hcl is selected as the API for its hydrophilic capabilities; making it water-soluble and reliably measured as it dissolves. Deposition of the API is then regulated by deposition through a piezoelectric nozzle.
Does it work?
After curing, the researchers measure the dissolution rate of the bioink Ropinirole Hcl pills in an acidic substance used to mimic stomach contents. Within the first 15 minutes of tests 60% of the tablet’s contents were released. After another 15 minutes, a further 20% was released, as shown in the graph below.
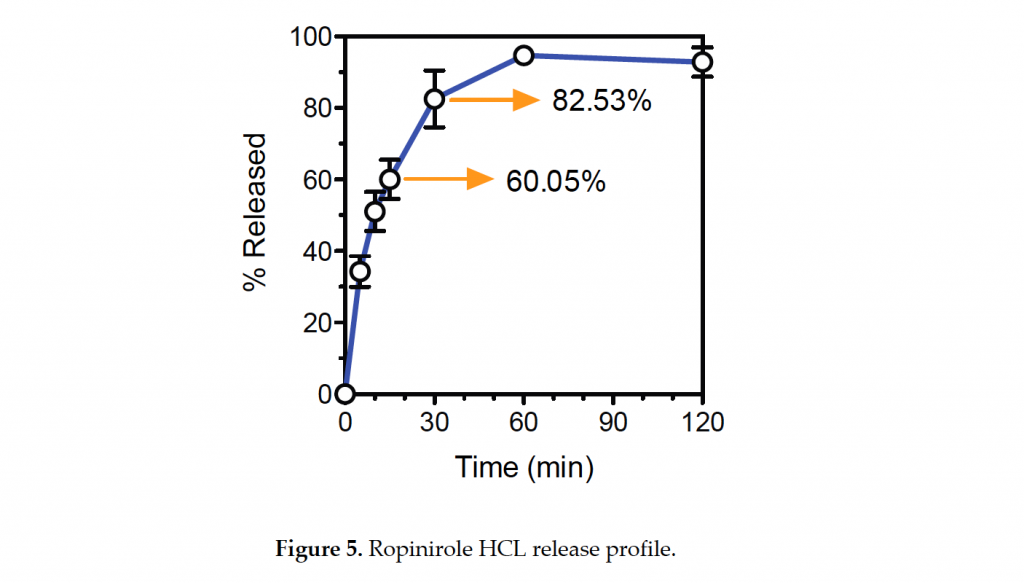
The peak of drug release is seen after an hour of dissolution, after which a small percentage (around 4%) of the contents are lost.
The future in PolyJet 3D printing
As a light-reactive ink, the Ropinirole Hcl hyaluronic acid becomes a potential candidate for use in PolyJet 3D printing. As of 2011 Stratasys own the patents to PolyJet technology which combines inject printing with photocuring to make 3D objects.
MIT scientist and artist Neri Oxman used PolyJet technology to produce the Wanderers: An Astrobiological Exploration collection of futuristic “wearable skins”.

Improving the translation between pharmaceutical substances and the PolyJet process could mean that tablets are entirely producible by 3D printing, rather than using prefabricated outer shells. Such a development would establish 3D printing as a viable method for pharmaceutical production.
These findings published in the Open Access Journal for Bioengineering from Swiss publishers MDPI (Multidisciplinary Digital Publishing Institute), are the collaborative effort of specialists from the Department of Chemical and Biomolecular Engineering, the Department of Bioengineering, and the Division of Advanced Prosthodontics and the Weintraub Center for Reconstructive Biotechnology.
We will be releasing our shortlist of nominees for the first annual 3D Priniting Industry Awards on 20th February. To ensure your company makes a spot, you can submit your nomination here.
Featured image shows a collection of colored pills, photo by v1ctor on flickr


