Researchers from the Federal University of Sao Paulo (UNIFESP) in Brazil have developed a new method to 3D print brain cells that are capable of surviving for at least 14 days after printing.
Funded by a research grant from the Sao Paulo Research Foundation (FAPESP), the study resulted in a model that is reportedly more similar to neural tissue than existing protocols, and which can be used to better understand the functionality of neural cells in relation to central nervous system diseases.
The researchers are also leveraging their new method to explore materials that could be used in the future to repair brain areas damaged by traumatic injuries or strokes.
“In the organism, cells are three-dimensional, but when grown in the laboratory they’re on top of plastic and underneath culture medium,” Marimelia Porcionatto, a professor at UNIFESP’s medical school and co-author of the paper, told FAPESP. “That’s very different from the natural organization of tissue or organs, where cells are arrayed in three dimensions.
“The bioink we developed attempts to reproduce the relationship between the cell and the microenvironment and other cells. It’s an intermediate system between 2D culture and experiments with animals.”
3D printing neural cells
Astrocytes are the most abundant cells in the nervous system and play an essential role in numerous neurological processes and in diseases that affect the brain. These particular cells form the basis of the researchers’ study, but the process they developed can also be adapted to study other cell types, too.
For instance, the team is currently using the method to analyze astrocytes and neurons infected by SARS-CoV-2, the virus that causes Covid-19, as part of another FAPESP-funded project.
“We’re testing different biomaterials for compatibility with neural tissue cells – neurons and neural stem cells, as well as astrocytes,” said Bruna Alice Gomes de Melo, first author of the study. “Bioprinting is a recent technique in tissue engineering, and neural tissue cells are particularly sensitive, so this protocol will be useful both for researchers working with astrocytes and other brain cells and for those working with other cell types.”
The method involves 3D printing a novel bioink laden with astrocytes to form a neural-like tissue structure, leveraging an extrusion-based 3D printer from 3D Biotechnology Solutions. The bioink itself is made up of astrocytes extracted from mice brain cortices and a solution composed of gelatin, laminin and other naturally-occurring biomaterials. Rather than importing gelatin methacryloyl (GelMA) from abroad, the team produced it themselves in the lab at a far lower cost.
“In other compositions, many of the cells survived the stress of 3D printing for a time, but astrocyte morphology wasn’t compatible with living tissue,” Melo added. “GelMA and laminin were essential.”
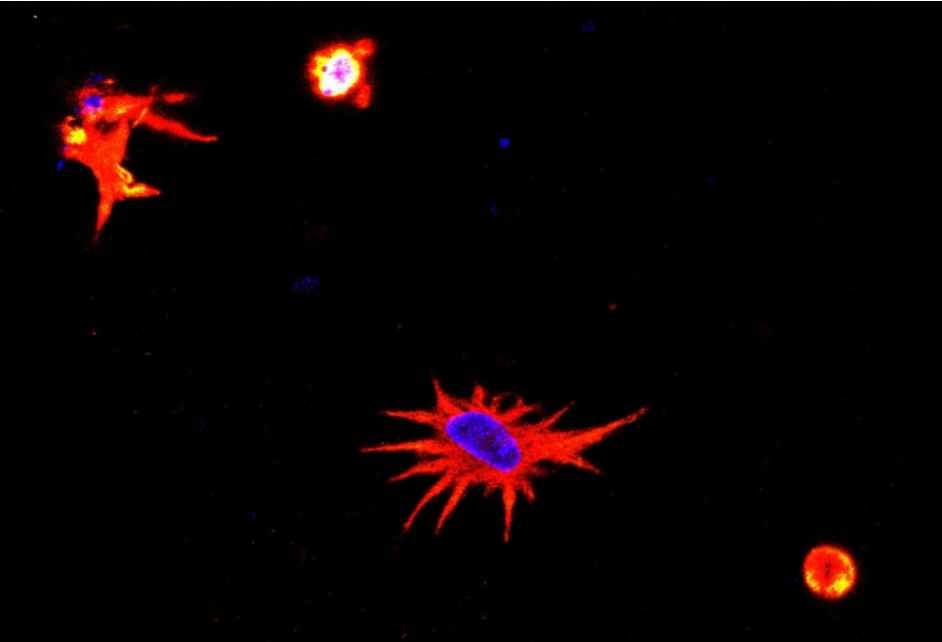
Fabricating “living” brain cells
A few days after being printed into structures, the astrocytes reportedly began to replicate and behave in a similar manner to that seen in nerve tissue. After one week of incubation, the viability of the astrocytes significantly increased, indicating that the 3D structure provided a suitable microenvironment for cell growth.
According to the researchers, the astrocytes were able to survive for at least 14 days after printing, providing a considerable time window to study their mechanisms and behavior.
Going forwards, the team plans to increase the complexity of its 3D printing method by including neural stem cells in the bioink mixture.
“The idea is to get as close as possible to the complexity of neural tissue,” Porcionatto told FAPESP. “When these protocols are fully validated with mouse cells, we’ll be able to develop others with human cells. They’ll be useful for a variety of studies, such as candidate drug trials, tests to identify genes expressed during brain development, and disease modeling, among others.”
Further information on the study can be found in the paper titled: “3D bioprinting of murine cortical astrocytes for engineering neural-like tissue,” published in the jove bioengineering journal. The study was co-authored by B. Melo, E. Cruz, T. Ribeiro, M. Mundim, and M. Porcionatto.

Advancing neurological treatments with AM
Bioprinting is receiving increasing attention for the opportunities it offers for the study of neurological conditions and potential new treatments. While the UNIFESP team’s study is novel in its ability to keep astrocytes alive for more than two weeks, there have been other significant breakthroughs in bioprinting brain cells over the past year.
Last January, medical tech firm Fluicell partnered with clinical R&D company Cellectricon and the Karolinska Institutet university to 3D print neural cells into complex patterns. The scientists were able to precisely arrange rat brain cells within 3D structures without damaging their viability, which could be used to model the progress of neurological diseases.
Meanwhile, the ongoing EU-funded Meso-Brain research project headed up by Aston University is developing 3D nanoprinting techniques to produce a new generation of accurate modeling and testing tools. The project partners hope to generate customizable 3D printed networks of neurons derived from stem cells which they believe could revolutionize the treatment of diseases like Parkinson’s and dementia.
In August last year, award-winning researchers from Tel Aviv University 3D printed a first-of-its-kind glioblastoma tumor using patient cells that could empower new methods to improve the treatment of brain cancer. Claimed to be the world’s first fully functioning 3D model of a glioblastoma, the customizable model enables the researchers to test the efficacy of new drugs in an environment that accurately mimics a patient’s individual tumor and brain.
Most recently, a team of researchers from the University of Montreal, Concordia University and the Federal University of Santa Carina successfully 3D printed living mouse brain cells using their newly developed Laser-Induced Side Transfer (LIST) 3D bioprinting technology. LIST reportedly overcomes the various limitations of other bioprinting techniques, such as donor preparation challenges, ink viscosity, and cell viability.
Subscribe to the 3D Printing Industry newsletter for the latest news in additive manufacturing. You can also stay connected by following us on Twitter and liking us on Facebook.
Looking for a career in additive manufacturing? Visit 3D Printing Jobs for a selection of roles in the industry.
Subscribe to our YouTube channel for the latest 3D printing video shorts, reviews and webinar replays.
Featured image shows morphology of a bioprinted astrocyte: cell nucleus stained blue and remainder stained red show a very similar state to that seen in neural tissue. Image via FAPESP.



