Ink-possible: that is what, playing with the word “ink”, most people, even many medical professionals, think of organ bioprinting. While it is true that the possibility of full complex organ digital manufacturing will become less “inkpossible” only a few decades from now, there are many aspects of bioprinting that are very real, now, and the biggest limitation they encounter is the confusion and lack of clear information about the products. Basically the same exact thing that happens in all 3D printing.
That’s because 3D bioprinting is not something separate from the rest of 3D printing. All digital additive manufacturing processes, including objects, food and organs, share a lot more than may seem at first. In fact they share just about everything and one Swedish start up called CELLINK has made that all the more clear, by focusing on one aspect that is fundamental in all printing: consumables.
CELLINK produces and sells a hydrogel that is compatible with all extrusion based 3D bioprintiners. Basically want to establish themselves as the leading suppliers of consumables as bioprinters become more widely available and their prices drop. While the founders business background brought them to identify consumables as the most strategic part of the future bioprinting industry, their vision is not limited to the business aspect.
“We have noticed this field is becoming very complicated and we don’t like that,” Erik Gatenholm, the young company’s co-founder and Director of Operations, tells me. “We want people to gain a better understanding of this technology and what we are doing. We feel that bioprinting is the future of how we are going to heal people, and it’s better to tackle the issues that surround it now, rather than when it will be a common practice in the clinics.”
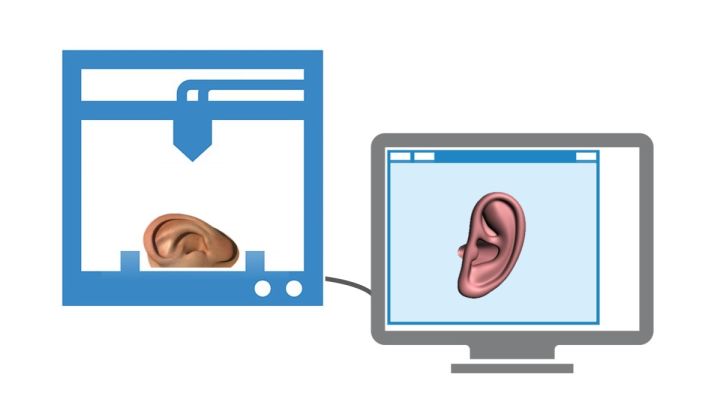
I must admit that when I first visited the CELLINK website, the first impression I got is that there were a lot of cool renders and that it was a good idea, with nothing really solid behind it. Browsing around the web I ran into CELLINK’s Facebook page where I was able to see that CELLINK is a product which is already being sold for research purposes and that has already been used to make very solid looking cartilage ears. After speaking with Erik and CELLINK’s other co-founder, Ivan Tournier, R&D engineer with a background in biotechnology, I have to say that everything they are doing – and bioprinting in general – is now, surprisingly, much clearer.
CELLINK is a bioink. It is compatible with most extrusion based bioprinters in the market and can be used to 3D print cartilagineous structures. “To be bioprintable a bioink needs to have two main characteristics,” says Ivan Tournier, “it needs to be biocompatible and it needs to be printable.” It seems like a basic concept however, especially when working with cartilage, reproducing the correct mechanical properties using a 3D printed material is the main challenge.”
Today most bioprinting is done by creating a scaffold using a biocompatible polymer. This can be PLA or another thermoplastic such as polycaprolactone. The scientists then apply the hydrogels, that is the cell cultures, onto the scaffold and cultivate them in order to make them reproduce and gradually fill the scaffold (which, ideally, will gradually dissolve away inside the body). Newer approaches such as the one implemented by CELLINK use a mixture of scaffolding material and cellular hydrogel into a single product, which can be bioprinted directly and thus used to make more complex shapes, faster.
“In the structures we have been able to print using our CELLINK we see that the cartilage cells begin reproducing by themselves and gradually take over”, Erik said. “This means that the final product’s mechanical properties will be virtually identical to natural structures and also that they could already be used for implantation in the body.”
What separates us from that is testing and institutional approval, which, considering we are at the very beginning of the process, is bound to require another 5 to 6 years. Nevertheless, CELLINK is already selling its consumables to universities and research institutions, which use it for drug testing and bioprinting experimentation in general. In Gothenburg, where they are based, they already work closely with the local Universities and orthopaedics who, as Erik put it, “have been very impressed by their progresses”.
The best part is that CELLINK is not stopping at cartilage. As professor Jos Malda from the Utrecht Bioprinting Center (which uses the RegenHU 3D printers, which have already been tested and used with the CELLINK) had explained to me last year, skin is the next frontier. “That is more challenging than cartilage as it has two types of cells,” says Ivan, “however we are already working on it and the first results are promising.”
What I find amazing about 3D printing is that small startups can innovate in fields that are traditionally dominated by giant corporations. The CELLINK team agreed that all 3D printing is part of the same, new and unique way to do things and that all aspects of it are so intertwined that discoveries in one can affect developments in another. By the same token, the elements that favour the evolution of all 3D printing are similar, starting from the need for clear and precise information, lowering cost of the machines, more advanced software and easy to use, quality materials. Erik said that their short term goal is to seek more funding to strengthen their company structure and distribution network. Perhaps the larger filament distributors might give it a thought. That would be ink-pressive