Researchers from Lund University in Sweden have developed a new 3D printable bioink that could bring the 3D printing of human organs one step closer to reality.
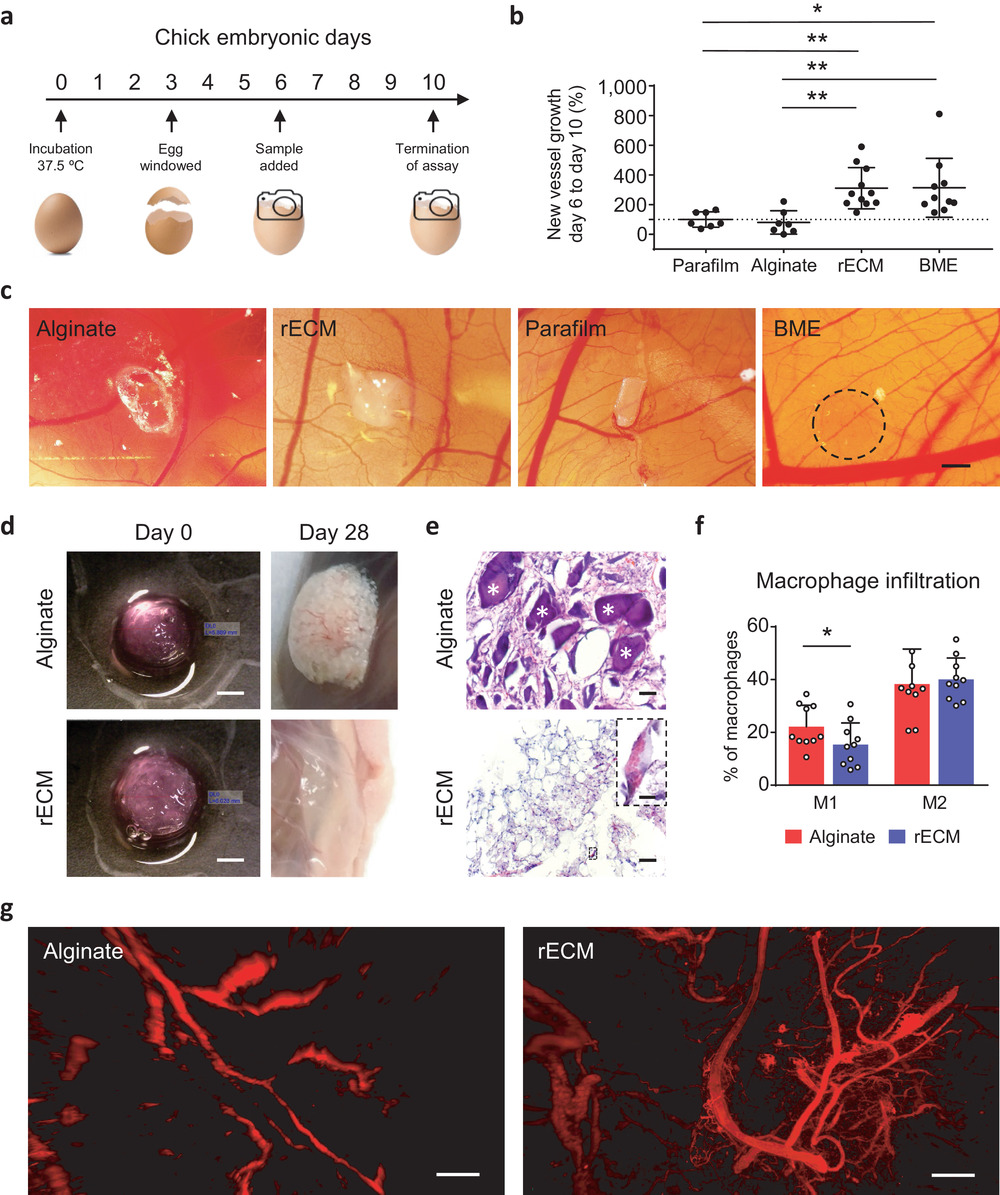
Made up of a combination of alginate derived from seaweed and lung tissue, the bioink enables biocompatible constructs that resemble human-sized airways to be 3D printed. Once printed, the constructs support new cell and blood vessel growth in the transplanted material.
While lung tissue was the focus of the initial study, the bioink can reportedly be adapted for any tissue or organ type.
As such, the researchers, led by Associate Professor and senior author of the study Darcy Wagner, believe their work identifies a promising new class of bioinks for 3D printing functional human tissue for transplantation.
3D bioprinting human tissue
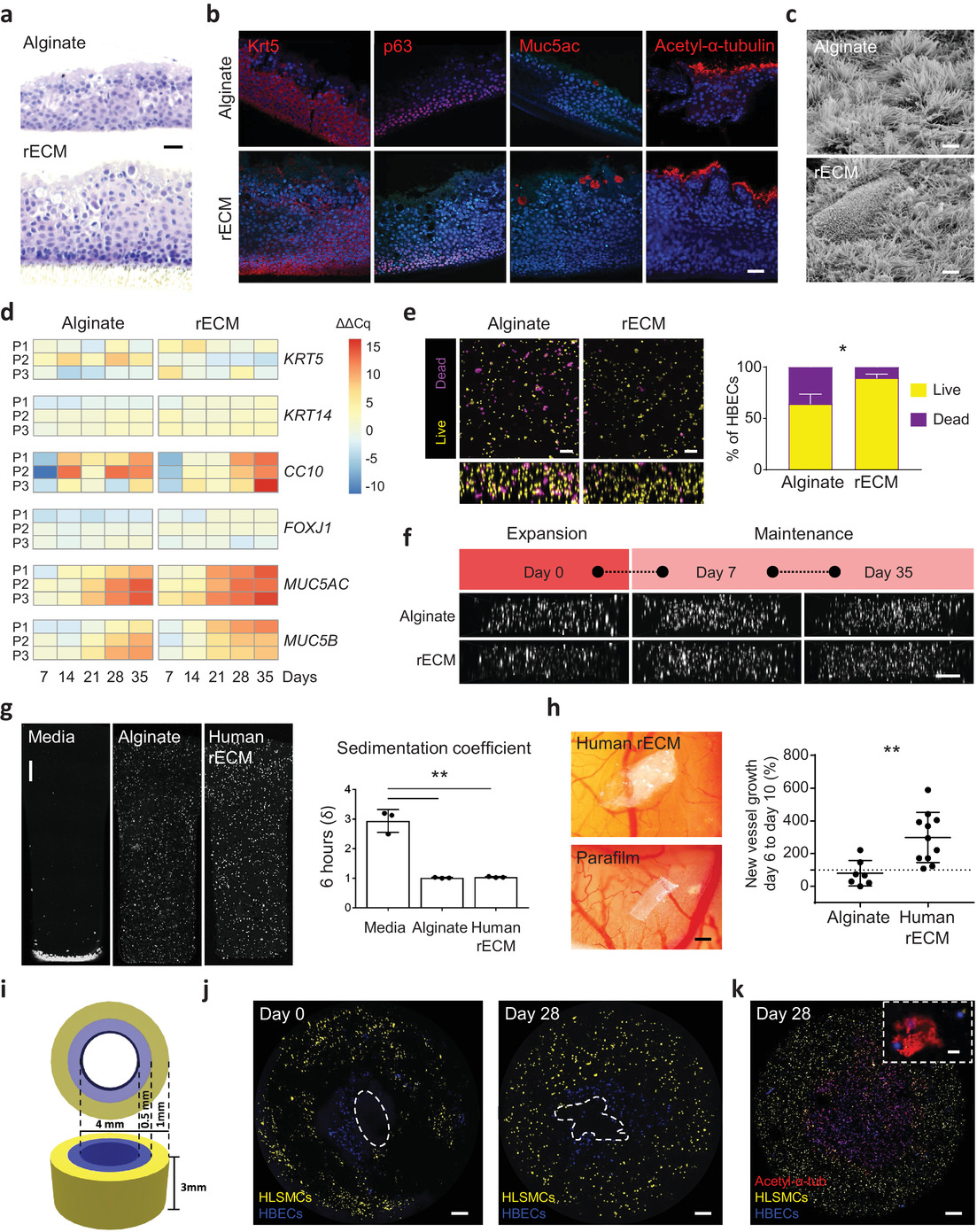
Wagner and her team began by combining alginate derived from seaweed with an extracellular matrix taken from lung tissue to form the bioink. The bioink was then laden with stem cells found in human airways and 3D printed to form complex and mechanically stable tissue constructs that mimicked these airways.
“We started small by fabricating small tubes, because this is a feature found in both airways and in the vasculature of the lung,” Wagner said. “By using our new bioink with stem cells isolated from patient airways, we were able to bioprint small airways which had multiple layers of cells and remained open over time.”
The 3D printed constructs included perfusable tubes and branching structures that spanned the anatomical length scales of human tissue, and which didn’t require external support structures. The presence of the extracellular matrix within the bioink helped to enhance the survival of human progenitor cells – descendants of stem cells that further differentiate to create specialized cell types – during the bioprinting process to enable tissue-specific cellular differentiation and vascularization of the implant within the transplant site.
For the transplant site, the team used a mouse model that closely resembled the immunosuppression used in patients undergoing organ transplantation. Immunosuppression refers to the state at which your immune system is not functioning as well as it should, which can be caused by medical procedures such as this.
According to Wagner, the 3D printed constructs formed from the developed bioink suppressed the foreign body response, were pro-angiogenic and supported blood vessel formation. This is a result of the bioink’s ability to maintain its biological activity both during and after the printing process.
“These next generation bioinks also support the maturation of the airway stem cells into multiple cell types found in adult human airways, which means that less cell types need to be printed, simplifying the nozzle numbers needed to print tissue made of multiple cell types,” she explained.
High-resolution 3D printing holds the answer
In order for Wagner and her team to continue advancing and improving their newly-developed bioink, the resolution of 3D bioprinting needs to be further improved. Higher resolution printing would allow the researchers to 3D print more distal lung tissue and alveoli, which are vital for gas exchange, and to bring fully 3D printed lungs closer to reality.
“We hope that further technological improvements of available 3D printers and further bioink advances will allow for bioprinting at a higher resolution in order to engineer larger tissues which could be used for transplantation in the future,” she said. “We still have a long way to go.”
Martina De Santis, first author of the study, added: “The development of this new bioink is a significant step forward, but it is important to further validate the functionality of the small airways over time and to explore the feasibility of this approach in large animal models.”
More information on the study can be found in the paper titled “Extracellular-Matric-Reinforced Bioinks for 3D Bioprinting Human Tissue”, published in the Advanced Materials journal. The study was co-authored by M. De Santis, H. Alsafadi, S. Tas, D. Bölükbas, S. Prithiviraj, I. Da Silva, M. Mittendorfer, C. Ota, J. Stegmayr, F. Daoud, M. Königshoff, K. Swärd, J. Wood, M. Tassieri, P. Bourgine, S. Lindstedt, S. Mohlin, and D. Wagner.
Advances in 3D bioprinting
While cell-laden bioprinted structures hold great potential for human tissue and organ transplant, the technology is still very much in its experimental stage. The development of suitable bioinks, limited print speeds, print resolution, and restrictions on architectural complexities are all hurdles to the technology’s wider adoption.
However, there have been several significant developments in the past year that show promise in advancing bioprinting and its applications.
Scientists from the University at Buffalo have recently developed a rapid new 3D bioprinting method that could bring fully-printed human organs closer to reality. The method reportedly enables centimeter-sized hydrogels to be printed between 10-50 times faster than is possible with conventional 3D printing techniques, and also enables the production of embedded blood vessel networks.
Meanwhile, US 3D printer OEM 3D Systems announced plans to ramp up its regenerative medicine and bioprinting activities, after experiencing a breakthrough in its Print to Perfusion bioprinting platform. The system is now capable of rapidly producing full-size, vascularized lung scaffolds and will play a key role in the firm’s healthcare business going forwards.
Elsewhere, biotechnology firm United Therapeutics and Israeli bioprinting firm CollPlant expanded their partnership for the mass-manufacture of 3D printed kidneys in September 2020, although the companies have since terminated their agreement in February this year. 3D bioprinting has been deployed to arrange neural brain cells into complex patterns that could be used to model the progress of neurological diseases.
Subscribe to the 3D Printing Industry newsletter for the latest news in additive manufacturing. You can also stay connected by following us on Twitter and liking us on Facebook.
Looking for a career in additive manufacturing? Visit 3D Printing Jobs for a selection of roles in the industry.
Featured image shows blood vessel infiltration in the 3D bioprinted constructs. Image via Lund University.



