Israeli regenerative medicine firm Matricelf has reached a new milestone in the development of its 3D printed neural implants for paralyzed patients with spinal cord injuries.
The firm has successfully produced its own in-house induced pluripotent stem cells (iPSCs) from human peripheral blood cells, which will be combined with a unique hydrogel to form 3D printed implants which could potentially cure paralysis.
“Our ability to independently produce human iPSCs is a huge achievement of the R&D team both in the scientific and business impacts,” said Asaf Toker, Matricelf’s CEO. “The ability to produce iPSCs without the need to rely on external sub contractures positions the company as a central player in cellular therapy and regenerative medicine industry and will reduce future manufacturing costs.
“We believe that independent manufacturing capacity of iPSCs is a remarkable economic opportunity in billions of USD market per year dealing with spinal cord injury, and in the future for a variety of other medical conditions.”
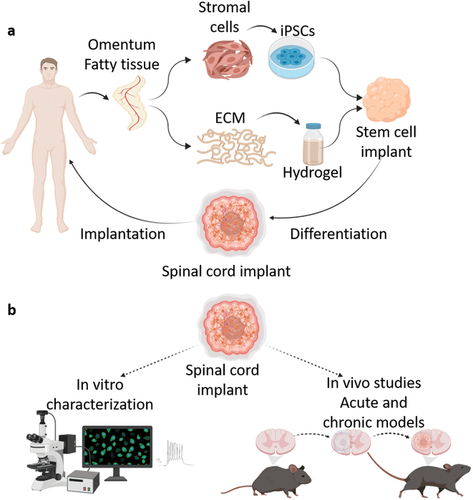
Matricelf’s 3D bioprinting technology
Founded in 2019, Matricelf leverages a patent-pending 3D bioprinting technology that has been under development at Tel Aviv University (TAU) for the last decade in the lab of Professor Tal Dvir, one of the founders of the Matricelf and the firm’s Chief Scientific Officer.
In January, Matricelf revealed it had signed an exclusive global licensing agreement with TAU’s technology transfer firm Ramot to use the technology, which was leveraged to produce what was reportedly the world’s first 3D printed heart back in 2019.
The bioprinting technology works by simultaneously 3D printing cells and extracellular matrix (ECM) derived from patients to produce living human tissues and organs. Liquid nano-particles stabilize the printed structures and provide high resolution and precision before being extracted after printing is complete.
In February, Matricelf announced its had successfully tested a “first of its kind” 3D printed spinal cord tissue implant on paralyzed mice that enabled them to walk again. The firm is currently gearing up to enter into human trials with its 3D printed spinal cord implants in 2024, which they believe could provide a potential cure for paralysis within just a few years.

Fabricating in-house human iPSCs
Matricelf’s latest milestone sees the firm able to produce in-house IPSCs from human peripheral blood cells for the first time. IPSCs are capable of differentiating into different cell types, including neural cells, and are widely used for therapeutic purposes such as regenerative medicine, disease modeling, and drug discovery.
By combining their self-developed iPSCs with a unique thermo-responsive hydrogel, Matricelf will produce 3D printed neural implants that could enable patients with spinal cord injuries to walk again.
The 3D printed implants are designed to enable the regeneration of damaged tissues within and around the spinal cord using a cellular and ECM component originating from individual patients.
Being able to self-manufacture iPSCs is a significant achievement for the company’s pre-clinical R&D program. According to the firm, independently producing iPSCs alongside the development of its hydrogel is a crucial step towards achieving end-to-end independent manufacturing capacity for its neural tissue implants.
“The process of reprogramming mature cells in iPSC, that present the potential to differentiate to any cell type, is a revolutionary and promising technology in the world of cellular therapy and regenerative medicine,” said Tamar Harel Adar, Matricelf’s VP R&D. “Independent manufacturing capabilities of iPSCs is a result of intensive work by the R&D team that is based on acquiring and developing applicable know-how and scientific tools.
“The ability to manufacture iPSCs from the patient’s own mature cells is significant since it enables the company to produce new tissues to replace various human damaged tissues in a variety of medical conditions.”
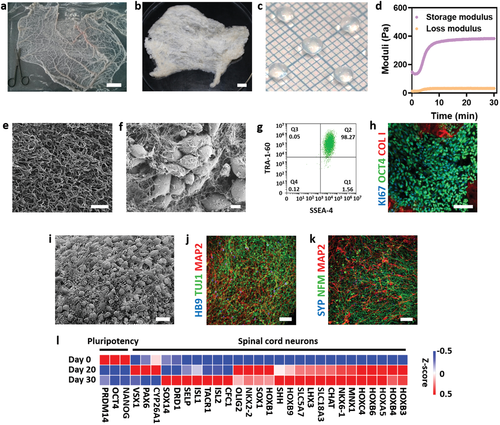
3D bioprinting holds great potential for enhancing the success rates of spinal surgeries and treatments, and Matricelf is not the only one making progress in this area.
In terms of research, the last few years have seen the development of 3D printed scaffolds containing living cells that could help to restore some function to patients with spinal cord injuries. Developed by the University of Minnesota, the bioprinted scaffold is comprised of silicone and stem cells and could form a ‘bridge’ between living nerve cells located around an injury. Meanwhile, scientists from the University of California San Diego have successfully 3D printed a two-millimeter spinal cord implant to repair spinal cord injuries in rats.
Regarding surgical applications, orthopedic implant manufacturer 4WEB Medical has launched its stand-alone 3D printed Anterior Spin Truss System designed to aid surgeons during spinal procedures, while 3D printed orthopedic device developer Orthofix Medical has unveiled its FORZA Ti PLIF Spacer System for use in posterior lumbar interbody fusion surgeries.
Subscribe to the 3D Printing Industry newsletter for the latest news in additive manufacturing. You can also stay connected by following us on Twitter and liking us on Facebook.
Looking for a career in additive manufacturing? Visit 3D Printing Jobs for a selection of roles in the industry.
Subscribe to our YouTube channel for the latest 3D printing video shorts, reviews, and webinar replays.
Featured image shows the stages of Matricelf’s 3D bioprinted spinal cord process. Image via Matricelf.



