3D printing software company Materialise (NASDAQ:MTLS), headquartered in Leuven, Belgium, has attained FDA clearance for surgical guides used to operate on forearm defects in children aged 7 and up. The devices allow for heightened precision in such delicate operations that typically only produce the planned effect in 40% of cases.
Bryan Crutchfield, Vice President and General Manager of Materialise North America, comments,
In bringing this 3D printing technology to pediatric surgery, surgeons will have access to our clinical engineers’ wealth of experience developing osteotomy guides, helping them perform even the most complex bone corrections that will have a positive impact on the rest of the child’s life.
An improvement on the existing procedure
A human forearm consists of two bones, the ulna (on the inner arm) and the radius (on the outer).
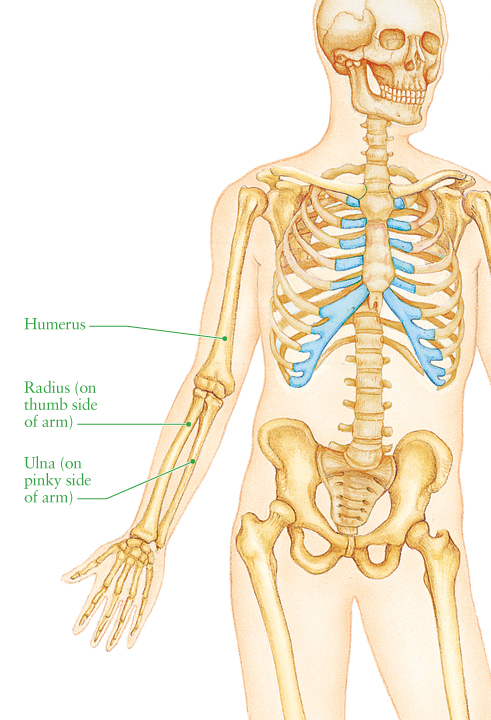
Discrepancies in the length of these bones, as a result of birth defects, diseases, or breakages, can cause immobility to the arm and further complications in later life.
In a clinical osteotomy of the forearm, bone is cut to allow realignment of radius and ulna bones, correcting the ability to move. Typically, surgeons create freehand surgical plans using x-ray images to plan the procedure, but 3D printing goes much further than this.
Giving children a better chance in life
In a case study published by Materialise in November 2016, a clinical osteotomy using 3D printing was performed on a 7-year-old boy, named Joos. Joss had previously fractured his arm, and when it healed the bones fused incorrectly, giving him a crooked arm.
By using arm x-rays and Joos’ healthy right arm as a model, hand specialist Dr. Frederik Verstreken was able to 3D print guides unique to Joos’ case.
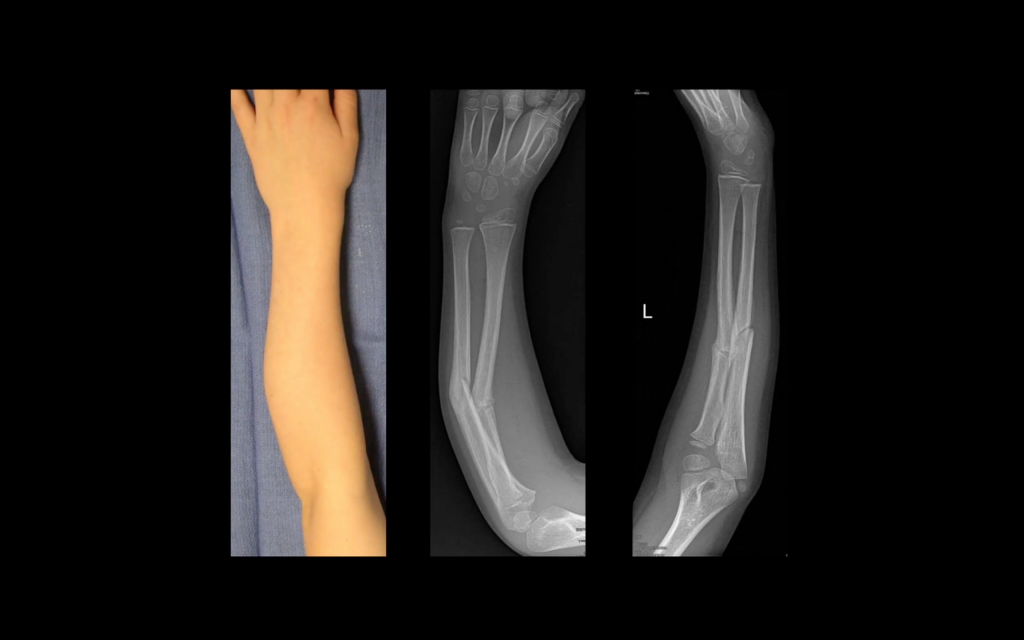
Materialise collaborated closely with Dr. Verstreken on the case, and after only a few days of healing, Joos regained feeling in his fingers. The boy has now regained full use of his left arm.
Prior to this development for paediatric care, Materialise built its expertise with over 1,000 clinical osteotomy consultations with adult patients.
The three Rs of 3D printing for medicine
In a video released in December 2016, the FDA shared its “3Rs of 3D printing” seeking to “Regulate, Research, and Resource” 3D printing methods in medicine, and develop reliable guidelines for the industry to follow.
Other 3D printed medical objects to achieve FDA approval include facial implants made by Oxford Performance Materials, and spinal support cages from the likes of Stryker. Spritam is the first 3D printed pill to be approved by the FDA, and was cleared in August 2015.
With obvious benefits to patient-specific medical devices, new objects are being passed all the time, accelerating towards a new standard within the industry.
To stay up to date on developments in 3D printing in medicine, sign up to the 3D Printing Industry newsletter, follow us on Twitter and like us on Facebook.
You can now vote for the best 3D printing applications in the first annual 3D Printing Industry Awards.
Featured image shows Joos van Dyck, a 7-year-old patient who underwent correctional surgery to his forearm with the help of 3D printing from Materialise. Image via Materialise on YouTube



