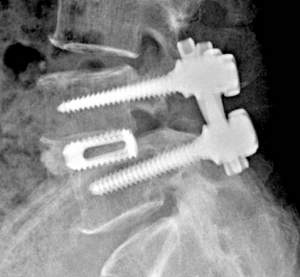
Stryker, a Michigan-based global leader in medical technology, has laser-sintered a Tritanium spinal cage for use in back surgery. The cages are hollow, and are used in-between vertebrae for patients with degenerative disc disease. The company only received FDA clearance for this design in March of this year, following a report from January revealing that Stryker had constructed a purpose-built factory for metal additive manufacturing.
The company started out life as part of Dr. Homer Stryker’s medical practice in Kalamazoo, Michigan. It was incorporated in 1946 as an orthopedic frame company, and upon Dr. Stryker’s retirement in 1964 changed its names to the Stryker Corporation, which is still based in Kalamazoo today. The Tritanium Posterior Lumbar (PL) Cage, is the product of a wealth of experience from Stryker corporation, and is going to be demonstrated at the North American Spine Society (NASS) Annual Meeting 2016 in Boston, from the 26th to the 29th October.
“We are excited to showcase the Tritanium PL Cage and unveil our new 3D virtual reality experience to surgeons during the NASS conference,” said Bradley Paddock, president of Stryker’s Spine division. “Oculus headsets will take surgeons on a journey through the evolution of Stryker’s pioneering additive manufacturing technology. They will then ‘travel’ to Stryker’s state-of-the-art additive manufacturing facility in Ireland for an up-close view of Tritanium and explore how our highly porous Tritanium implants may benefit their patients.”
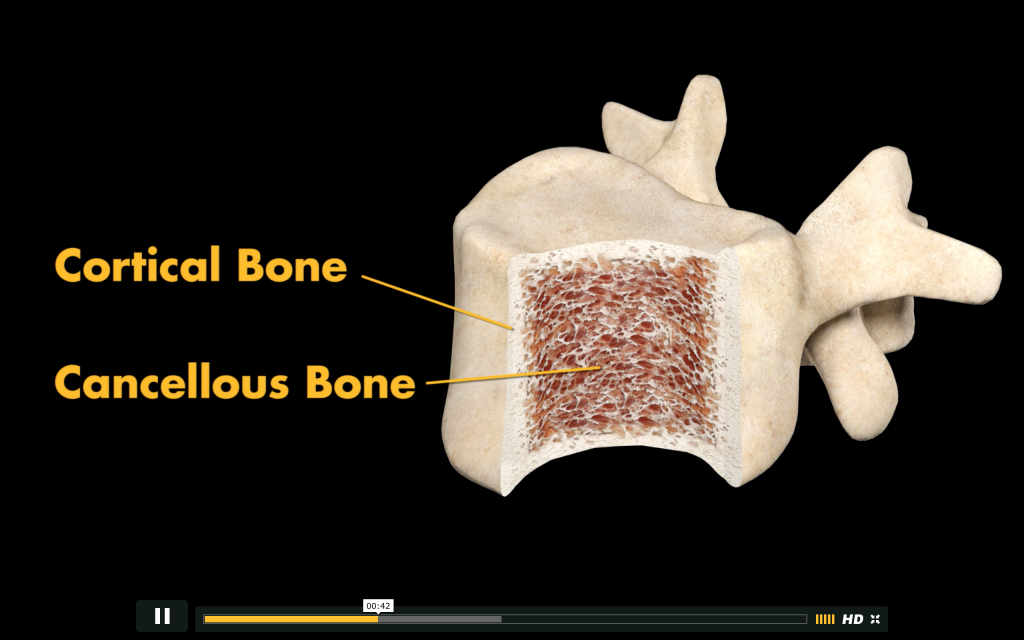
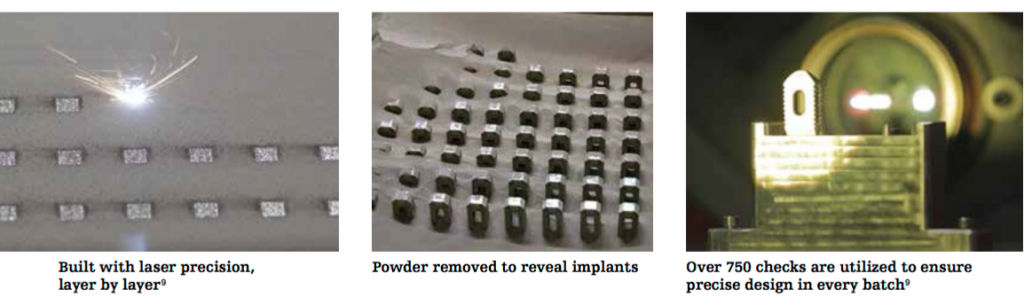
In pop culture, (and for all you Star Trek and Transformers fans out there) Tritanium is referenced as a material ‘21.4 times harder than diamond’. In reality, Tritanium is the brand name of Stryker’s titanium alloy material and used in their metal powder bed laser sintered 3D printing process. Making a Posterior Lumbar Cage in such a way allows the material to meet the same pore-structure and porosity specifications as naturally found in the human bone that it is made to replace. As seen with the manufacture of hip actuator cups, these pores can encourage bone in-growth improving longevity of the surgical implant. The 3D printer can make both the dense, cortical structure of a bone that allows the fusion of blood vessels, and the spongy, cancellous outer shell, often where the production of blood cells takes place.
Typically lumbar cages are often made of titanium, but in recent studies, such as the Percutaneous Transforaminal Lumbar Interbody Fusion (pTLIF) with a Posterolateral Approach for the Treatment of Degenerative Disk Disease: Feasibility and Preliminary Results, by Rudolf and Christian Morgenstern, published in July 2015, researchers have been exploring the suitability of the high-grade plastic PEEK for use as Posterior Lumbar Cages. In addition to the Oculus 3D demonstration, Stryker will also be presenting its results from a study entitled “Biomechanical and Histologic Comparison of a Novel 3D-Printed Porous Titanium Interbody Cage to PEEK”.
This study has gathered findings from a series of pre-clinical animal trials exploring the biomechanical performance and bone in-growth potential of various lumbar interbody fusion implants utilizing different materials, including the Tritanium PL Cage.

It will be interesting then, to see how a Tritanium PL Cage compares to competing materials, and not just how a living body reacts to the implant of foreign matter, but how it enhances recovery, as that is one of the many problems challenged by research of this kind.
Featured image: the Tritanium Lumbar Cage by Stryker via, Stryker.com



