Researchers from the City University of Hong Kong have created 3D printed microrobot carriers which transport cells within living organisms (in vivo) for targeted therapy and tissue regeneration.
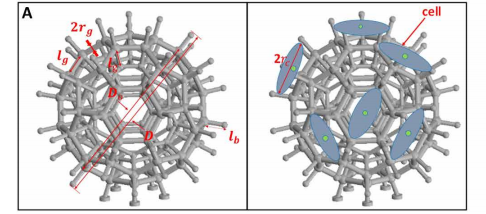
The spherical and barbed structure of the robots were fabricated using a Photonic Professional GT laser lithography system from Nanoscribe, which provided “sufficient flexibility to optimize the robot structure,” according to the research paper.
Cell regeneration through 3D printed Microrobot carriers
Regenerative medicine uses healthy cells from an organism (animals, plants, or humans) to repair or replace diseased cells or tissues. However, challenges arise when transporting functional cells to a damaged location within an organism. The study states:
“The delivery of [stem] cells in vivo requires a suitable three-dimensional (3D) structure that creates an environment that supports cell adhesion, proliferation, and differentiation while functioning as a carrier.”
Thus, the research team created several 3D printed microrobot carriers with magnetic and porous properties to mechanically support tissue and organ in situ regeneration. Furthermore, the researchers observed that a 2D cell-culturing artificial environment would be ineffective as it quickly loses its shape.
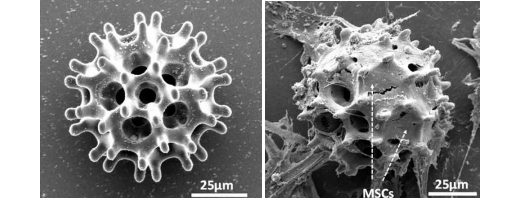
With a minimally invasive design, the microrobot carriers have the potential to access smaller and more complex regions of the human body. This includes gastrointestinal organs, the brain, and the spinal cord. Considering this, the research used Nanoscribes’ two-photon lithography technology which is capable of high-resolution 3D patterning through photonic crystals. The microbot carries were fabricated from a negative photoresist SU-8 50 material.
In addition, the microrobots were coated with nickel and titanium solutions for magnetic actuation and biocompatibility.
The mouse and the zebrafish embryo
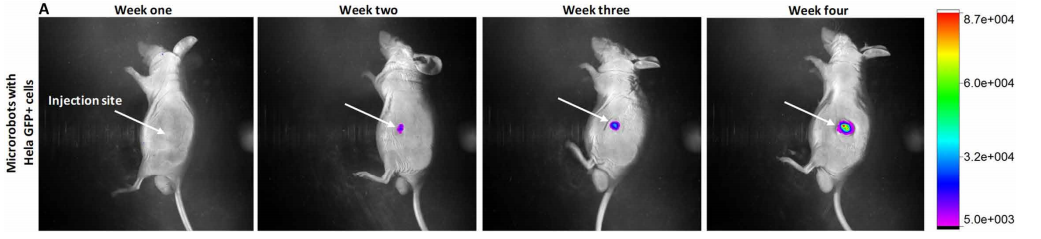
To test its cell-controlling and delivery capabilities, the research team dispersed a swarm of microrobots carrying HeLa protein cells, capable of generating tumors, into a mouse. Following four weeks of cultivation, the mouse developed tumors in the surrounding location of the injected microbot carriers.
Moreover, microrobot carriers were dispersed into the yolks of zebrafish embryos to observe injection accuracy and its ability to overcome viscous resistance.
Both tests were observed under a microscope and were deemed successful for precise, automatic cell transportation. Concluding the experimentation phase, the researchers found that the spherical 3D printed structure of the microbot carriers “enhanced the magnetic driving capability allowing easy fusion of the microrobot with host tissues and facilitating cell transfer from the robot to tissues.”
The researcher paper “Development of a magnetic microrobot for carrying and delivering targeted cells” is co-authored by Junyang Li, Xiaojian Li, Tao Luo, Tao Luo, Chichi Liu, Shuxun Chen, Dongfang Li, Jianbo Yue, Shuk-han Cheng, and D. Sun.
For more of the latest 3D printing released research news sign up to the 3D Printing Industry newsletter, Also, follow us on Twitter, and like us on Facebook.
On the lookout for new talent or seeking a career change? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows SEM images of the 3D printed microrobot carrier cultivating cells over a period of time. Image via Science Robotics/City University Hong Kong.


