Though it takes just an instant for research to become a discovery, an apple falling from a tree; a bolt of lighting; the snap of a shutter, fully developed breakthroughs in science and medicine take years to perfect, and what we read today, about Harvard material scientists’ developments in the bioprinting of a kidney structure, is one small step closer to a future where organ transplant lists are a thing of the past.
A paper published on 11th October 2016, entitled Bio-printing of 3D Convoluted Renal Proximal Tubules on Perfusable Chips, illustrates how 3D bio-printing has been used to create ‘proximal tubules’ (PTs) – minute vessels found within the structure of human kidneys, as pictured below:
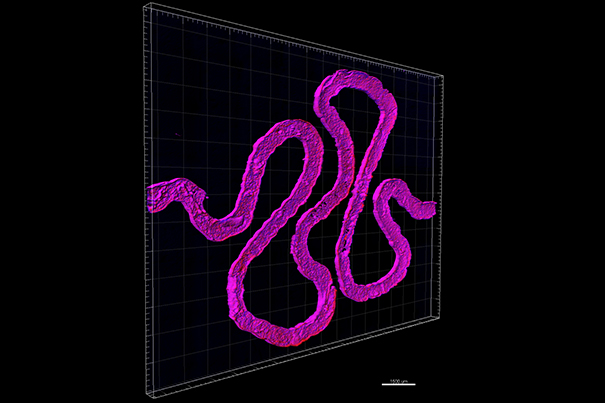
A proximal tube (PT) is a smaller segment of a nephron – which make up the structure of a kidney in their millions. Nephrons are what enables a kidney to filter and absorb nutrients from the blood.
How is it done?
Step 1: A PT structure is printed onto a glass slide (and it looks a lot like London’s River Thames)
Step 2: Then an extracellular matrix, matter that surrounds cells within the body, is injected on top of the river-like shape
Step 3: After this, the printed ‘river’ is removed to leave a hollow mold
Step 4: Living cells are injected into the mold which acts as a guideline for the structure of a proximal tube
Step 5: As the cells grow – or culture – they take on the characteristics of native PTs and can be tested with substances such as new medicines.
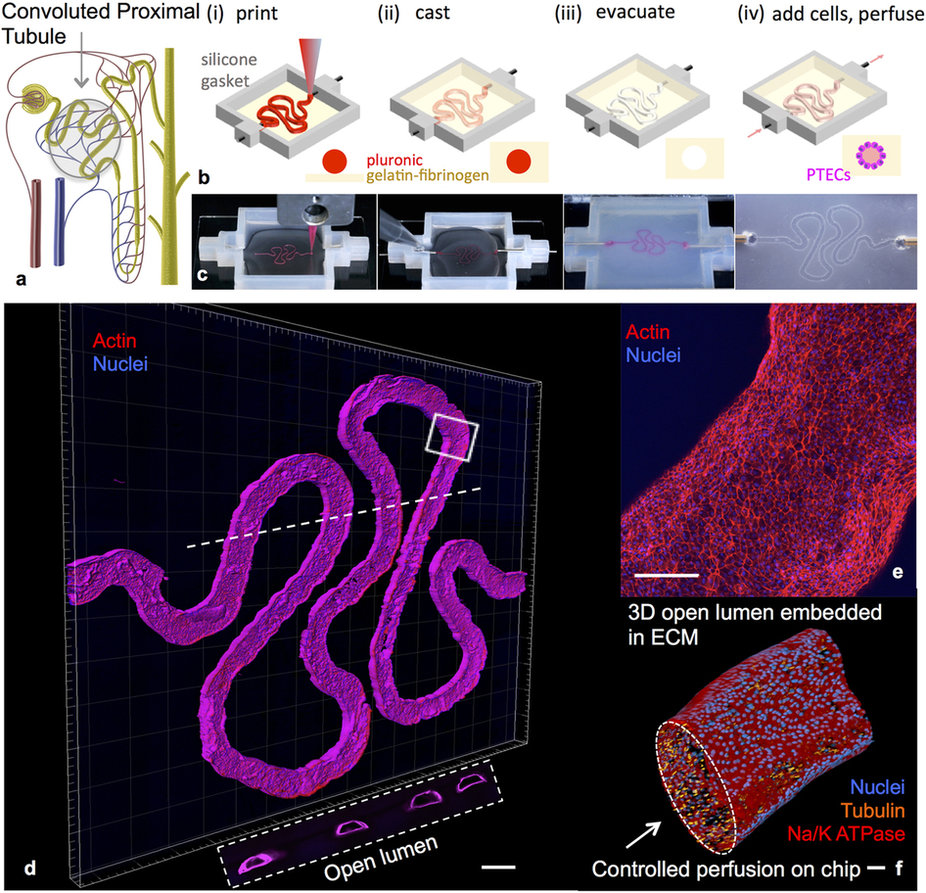
The process follows the same ‘sorting-out’ concept of cell manipulation that has been used in bio-printing a miniature heart and kidneys. The process of sorting-out means that cells given a particular form eventually retain the order of the synthetic structure around them, and operate as they would in this structure within the body.
Jennifer Lewis is the lead researcher on the project. As a professor at Harvard University, and core faculty member at the Wyss Institute for Biologically Inspired Engineering, Lewis is one of the leading names in 3D bio-printing research, and has over 120 journal articles attributed to her name. Her team at Harvard also succeeded in printing thick tissues containing blood vessels earlier this year, this research served as a basis for this most recent advance.
The team’s next step is to arrange the PTs into a vascular structure, gradually building up tissue until it is suitable to be transplanted. As the researchers write in the paper,
Three-dimensional models of kidney tissue that recapitulate human responses are needed for drug screening, disease modeling, and, ultimately, kidney organ engineering. Our bioprinting method provides a new route for programmably fabricating advanced human kidney tissue models on demand.



