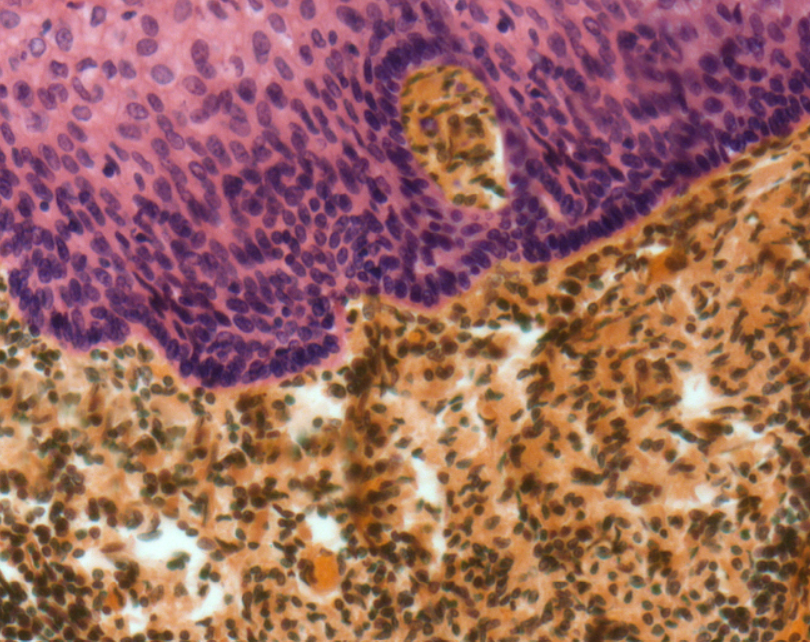
Researchers at Amgen British Columbia (BC) are using human tonsil tissue and 3D bioprinting to create an immune system model that can be studied outside of the body.
The goal of the study is to make a platform that can predict immune responses to new drugs more quickly and accurately. The researchers discovered that human tonsil tissue can be cultured in the lab to form 3D clusters that act identically to immune cells in the body. The platform has already been tested with substances that are known to either trigger or avoid immune responses. In both cases, the cells worked normally.

Tonsil organoids expand customizable immune tissue platform for drug development
The researchers intend to expand the tonsil organoid platform by constructing an in-house biobank of tonsil tissue. This will enable them to examine a more diverse population and gain a better understanding of a wide range of immune responses to drugs developed by Amgen. In addition, the team has partnered with FluidForm, a company that specializes in human tissue bioprinting technology, to develop immune tissue. The researchers aim to use engineered molecules and cells as building blocks to create the tissue and 3D bioprint it using FluidForm’s FRESH platform. This approach allows for the customization or engineering of all the necessary components, providing greater control over various immune responses.
Predicting immune responses to protein-based medicines is a significant challenge during drug development. The newly developed immune tissue platform has the potential to assess if a drug in development will trigger an immune response before it reaches clinical trials. This in turn will reduce the risk of trial failure. The team believes this platform could aid in the development of new antibodies.
Amgen’s clinical immunology group is working closely with the immune tissue project to evaluate the risk of immune responses to drugs in development. The conventional platforms used for assessment are limited in predicting the causes of immune responses once the drug reaches the clinic. The immune tissue platform has the potential to reduce the likelihood of clinical trial failure due to drug-induced immune responses.
Drug testing made easier with 3D printing
Researchers from the University of Stuttgart and Robert Bosch Hospital developed a 3D printed tissue platform that provides the possibility of replacing animal testing in clinical trials. The project received €3.8 million in funding from the German state of Baden-Württemberg. The project aimed to create an ex-vivo tissue model for evaluating the efficacy of cancer drugs. The joint team hoped to create skin-like microfluidic structures using a 3D bioprinter and simulation data to provide more correct estimates of tumor progression.
A team of researchers from the Wake Forest Institute for Regenerative Medicine (WFIRM) in North Carolina concluded the 3D bioprinting of a microscopic model of the human body. This laboratory model includes most of the vital organs and will be utilized to identify potential adverse effects of drugs before they are tested on humans. The experts anticipate that this sophisticated lab model will play a critical role in accelerating the drug development process, minimizing the expenses involved in clinical trials, and decreasing the need for animal testing.
What does the future of 3D printing for the next ten years hold?
What engineering challenges will need to be tackled in the additive manufacturing sector in the coming decade?
To stay up to date with the latest 3D printing news, don’t forget to subscribe to the 3D Printing Industry newsletter or follow us on Twitter, or like our page on Facebook.
While you’re here, why not subscribe to our Youtube channel? Featuring discussion, debriefs, video shorts, and webinar replays.
Are you looking for a job in the additive manufacturing industry? Visit 3D Printing Jobs for a selection of roles in the industry.
Featured image shows Light micrograph of a section through the surface of a tonsil showing the epithelium (pink) and underlying lymphoid tissue (brown). Image via Amgen.



