In previous years, SXSW Interactive has maintained health and medical technology programming, but this is the first year that the SX Health and MedTech Expo has existed in its expanded form. SXSW set up a whole slate of programming in this genre at JW Marriot, one of Austin’s shiniest hotels.
I was fortunate enough to attend “3D Bioprinting: The Next Revolution in Healthcare“, a discussion presented by Laura Bosworth, CEO and co-founder of TeVido BioDevices, and CTO Dr. Scott Collins. This particular session was quite full and eventually had to turn people away, which indicates that there is high interest in the topic. Hype is common in this environment, but the potential ramifications that 3D printing can have on the medical field are genuine. The hype here might be justified.
Lately, there has been much buzz about the possibility of 3D printed organs. Organ shortage is a big issue. An average of 21 people die each day on the organ donation waiting list. The need is real, but the reality is that simply printing out a new heart, lung, or kidney is still a far ways away.
The TeVido session presented some of what actually has been done in the field. There have been 3D printed ears, spine inserts, an aortic graft, and, even, craniofacial transplants. In many cases, the transition from academia to clinical use is already beginning. Recently the University of Michigan 3D printed a trachea, from dissolvable sutures, resulting in the saving of a infant’s life. This was described as an “exception” in the bioprinting and medical industries, due to the fact that the infant’s condition was immediate and life-threatening.
With living tissues, there are a number of additional issues that come into play, making the whole process even more complicated. Living cells, for instance, require a network of capillaries to provide blood flow, a huge hurdle to overcome. There is also a huge challenge that comes from the importance of structural integrity and the fidelity of placement. And, for organs like the kidney or liver, there is a hierarchical structure that must exist for a printed organ to function.
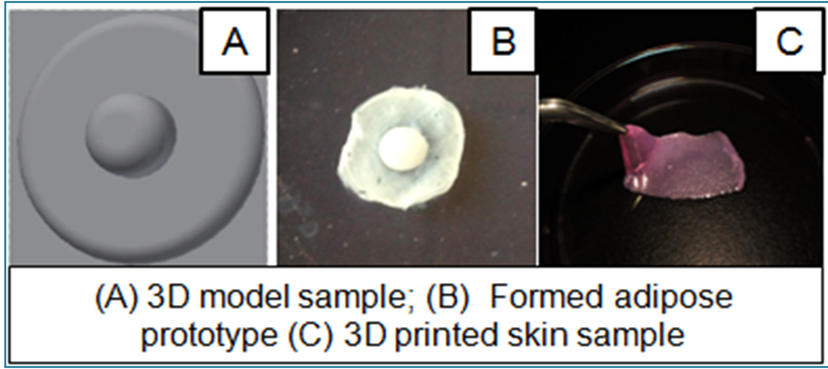
That is why TeVido Biodevices suggests addressing an unmet need in medicine that involves relatively small tissue and a relatively simple cell. As opposed to hearts and livers, which are quite complex, they propose to begin with 3D printed nipples. Current practices for post-mastectomy reconstruction of nipples are someone archaic: physicians make cuts in the skin, pinch it together, and let it heal for a few months. After that, a tattoo is applied to the scarred tissue to reconstruct the illusion of the previous form. Unfortunately, flattening and fading of this reconstructed form are considered the norm, and are, thus, only temporary.
The TeVido method, however, involves pre-surgical imaging of a patient’s nipple to create an accurate model for a bioprinted nipple. Then, a biopsy would capture a bit of the patient’s very own fat cells with which to 3D print a nipple based off of this 3D model, using a modified HP, inkjet printer. The usage of adult-differentiated tissue reduces the possible carcinogenic risks that come with stem cells (namely the cells going rogue and making more cancer) and, because the overall bioprint would be little more than a centimeter in diameter, it wouldn’t require surgeons to connect veins for blood flow.
Capillaries are adaptive and would expand to the area based on need, connecting naturally to the tissue and allowing the body to do most of the legwork. Moreover, the usage of the patient’s own cells eliminates the need for the potential hazards involved in anti-rejection meds. Because the tissue is truly part of the patient, it can potentially grow and change with them through their lives, a more accurate and genuine reconstruction than is available with current methods. Not only are vascular systems not required for tissue the scale that TeVido is working with, but the firm is also working with an inkjet system, rather than an extrusion system. According to the firm, their inkjet-based system has “higher fidelity and resolution” than the syringe-based extrusion machines implemented by other firms.
Currently, the company is performing research to ready their product for FDA approval. To do so, the company is hoping to pass beyond the prototyping phases and ready a final product, discussing the technology with plastic surgeons, doctors to make the printer, easier, faster, and cheaper. But, before they can have their process out of the lab and into the clinic, TeVido must perform numerous clinical trials and Collins states that it may be three years before their technology even sees the public.
1.4 million women around the world are diagnosed with breast cancer every year and, with early detection more frequent and fatality less common, TeVido is working to improve the quality of life of cancer survivors. This cause alone is a noble one, but funding this research would drive their technology even further, leading to potential breakthroughs in other bioprinted tissues, as well. What is learned and improved in this process could go on to help other work in bioprinting reach the next level of innovation.