Connecticut-based 3D bioprinting firm A.D.A.M. (Advanced Development of Additive Manufacturing) has received 510 (K) clearance from the U.S. Food and Drug Administration (FDA) for its 3D printed bioceramic and modified biopolymer bone implants.
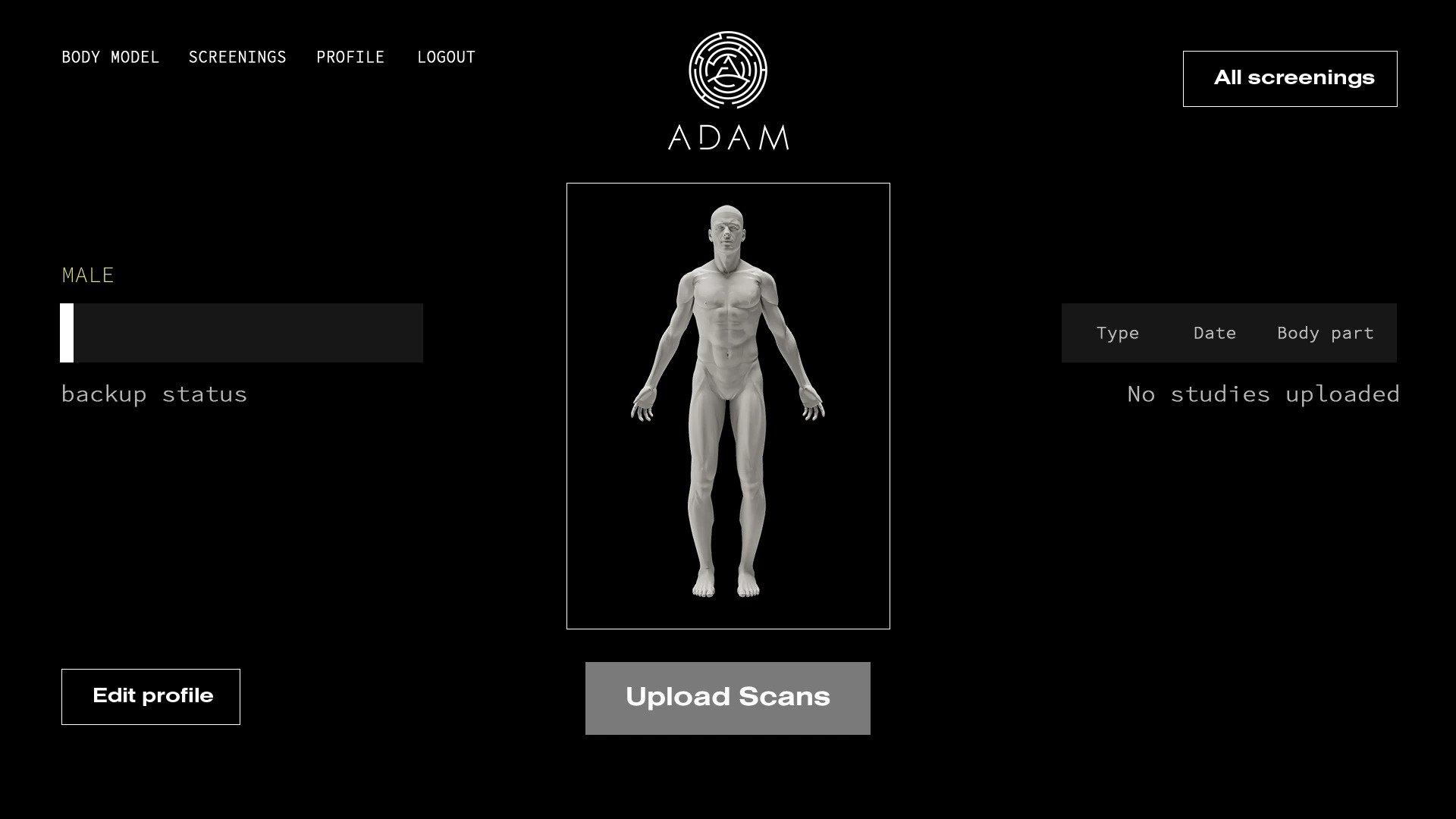
A.D.A.M.’s vision is to eventually enable patients to have their tissues modeled using MRI and CAT scans stored on its A.D.A.M. digital platform, which can then be 3D printed and implanted in designated clinics.
“The FDA’s response means a significant reduction in the time to market the product, from five years to one and a half years, since only the animal tests are required,” said Denys Gurak, A.D.A.M. CEO. “We are elated to be able to deliver a new breakthrough treatment standard for patients much sooner than previously expected.”
Printing body parts on demand
According to data from A.D.A.M., 150,000 people have died while waiting for an organ transplant in the last 20 years. This issue is one of the driving forces behind the company, which states “the only way to fix this is to start creating human organs and bones on demand.”
The project was initiated in August 2018 and entered a seed funding round in December that year. The following February, A.D.A.M. set up its laboratory, and a couple of months later started the development of its quality management system (QMS), which received certification in October 2019. Just over a year ago, the firm kickstarted the development of its digital platform. Now that A.D.A.M. has received 510 (K) eligibility from the FDA, the first step of bringing its vision to market is nearly complete.
A.D.A.M.’s business model aims to sidestep the time-heavy bone graft and bone donor search process, as doctors would instead be able to scan the area and 3D print the required bone through a combination of binder jetting and extrusion bioprinting techniques. Once printed, the bone can then be implanted into a patient, and ideally, dissolve over time as new bone grows.
The bones will be constructed from A.D.A.M.’s own modified biopolymer and ceramic bio-glass materials, which hold strong biocompatibility, feasibility, and structural properties when compared to naturally-formed bones.


A.D.A.M.’s long term strategy
With animal trials due to start imminently, A.D.A.M. will move its research and development (R&D) office to the University of Connecticut (UConn) as part of the Technology Incubation Program (TIP). Through this, the company will gain access to UConn’s advanced research facilities and business support services, in addition to its students.
A.D.A.M. isn’t just focused on bones, though. The firm’s long term strategy is guided by a self-proclaimed “transhumanistic vision” which will see other tissues 3D printed using its proprietary technology.
“Our plan is to go beyond bones and allow people to have their tissues and organs printed for an affordable cost, when and where they need them,” Gurak added. “This will allow millions to live their lives to the fullest. Concurrently, our R&D team is looking at potential uses for polymers in printing other tissues, such as heart valves and blood vessels. The horizon is bright.”
3D bioprinting
Realistically, the prospect of fully functional 3D printed transplant organs and tissues is still several years away. However, additive manufacturing has been utilized in several promising developments with future potential applications within regenerative medicine over the last few years.
In July, researchers from the University of Minnesota developed a novel bio-ink enabling them to create a functional 3D printed beating human heart, while elsewhere scientists from Tsinghua University 3D bioprinted brain-like tissue structures capable of nurturing neural cells.
Most recently, a global group of researchers developed novel nanoclay-based 3D bioprinted scaffolds with the potential to be used in skeletal regeneration. The implantable nanocomposite scaffolds were laden with cells derived from human bone marrow and human umbilical vein, which have the potential to facilitate bone formation.
Bioprinting has also taken off in space. In May, microdispensing specialist nScrypt and aerospace company TechShot successfully completed the first functional 3D bioprinting experiment in space of a human knee meniscus. Prior to this, 3D Bioprinting Solutions, a Russian bio-technical research laboratory, 3D bioprinted bone tissue in zero gravity on the International Space Station (ISS).

Subscribe to the 3D Printing Industry newsletter for the latest news in additive manufacturing. You can also stay connected by following us on Twitter and liking us on Facebook.
Be sure to subscribe to the Another Dimension podcast on your chosen podcast player to make sure you never miss an episode.
Looking for a career in additive manufacturing? Visit 3D Printing Jobs for a selection of roles in the industry.
Featured image shows A.D.A.M.’s digital scan platform. Image via A.D.A.M.



