A 3D printed bone graft from medical device manufacturer Cerhum has been given approval for use in patients throughout Europe.
MyBone is reportedly the first commercially available 3D printed bone graft authorized under the Medical Device Regulation 2017/745, and has also been ISO 13485 certified. As a result, the patient-specific bone graft will now be made available to maxillofacial and orthopedic surgeons throughout the continent.
“We are very proud to have successfully gathered vital clinical data and passed all the regulatory hurdles with a synthetic bone graft that is safe and effective,” said Grégory Nolens, Cerhum Founder and CSO.
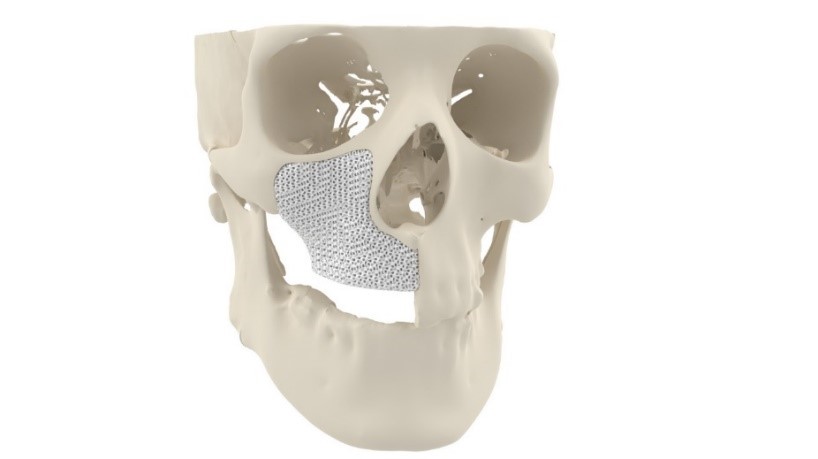
3D printing maxillofacial implants
Additive manufacturing is being increasingly leveraged for maxillofacial surgeries in the form of guides that enhance patient outcomes, and more recently for the creation of facial grafts that can be implanted into patients’ skulls.
For instance, the Québec Industrial Research Centre (CRIQ) is 3D printing patient-specific lower jawbone implants, while researchers at Paulista University have 3D printed a facial prosthetic for a Brazilian cancer survivor. Meanwhile, earlier this year, Health Canada approved its first Canadian-made 3D printed medical implant for use in facial reconstruction surgery for patients with oral cancer.
In May, a group of Korean researchers carried out a retrospective study to verify the effectiveness and safety of 3D printed titanium implants on maxillofacial bones. The study observed the outcomes of 16 patients with various maxillofacial defects, with almost all of the implants showing satisfactory treatment outcomes.
The MyBone bone graft
Cerhum (Ceramics for Humans) was founded in 2016 as a spinout from the Belgian national science and technology institute, Sirris. Partnered with the University of Liège, the company’s goal is to replace autografts – bone taken from elsewhere in the patient’s body – with its synthetic 3D printed alternative.
To date, the firm has 3D printed tens of thousands of synthetic bone grafts that have been used by a range of companies within orthopedic implants.
MyBone is a patient-specific 3D printed bone graft designed to treat patients with severe facial deformities. The graft is made of hydroxyapatite, a calcium phosphate that is the main mineral component of natural bone, and is 3D printed with a unique porous structure to encourage bone growth.
“Our 3D printed bone implants offer a unique, patented porous structure that allows ingrowth of blood vessels,” said Nolens. “This process, called vascularization, is key to achieving successful bone ingrowth. As a result, MyBone has shown seven times faster bone ingrowth than currently available bone graft granules.”
MyBone has now been validated for use in European patients under the Medical Device Regulation 2017/745 and been registered with the Belgian Competent Authority. The bone graft is also ISO 13485 certified, and as such is now available to surgeons as a patient-specific implant for maxillofacial surgeries.
As part of a controlled release phase two years ago, a patient received a MyBone graft implant which now reportedly looks exactly like natural bone according to a recent CT scan.
“Given the complexity of the defect, it would have been impossible to achieve such perfection from an aesthetic point of view and such a functional result with current methods,” explained Dr. Christophe Ronsmans, Head of the plastic surgery department at CHR Liege.
The patient is said to be doing “excellently” and the success of the surgery has helped pave the way for MyBone’s commercial availability in Europe.
“Due to extremely positive feedback of maxillofacial surgeons so far, Cerhum is expanding its portfolio into the dental and oncological market,” Nolens added.
Subscribe to the 3D Printing Industry newsletter for the latest news in additive manufacturing. You can also stay connected by following us on Twitter and liking us on Facebook.
Looking for a career in additive manufacturing? Visit 3D Printing Jobs for a selection of roles in the industry.
Subscribe to our YouTube channel for the latest 3D printing video shorts, reviews, and webinar replays.
Featured image shows Cerhum’s MyBone 3D printed facial bone graft. Image via Cerhum.



