3D printed medical implants and healthcare services are finding increasing use by healthcare professionals. While the number of cases is increasing, 3D Systems alone has provided services in over 100,000 cases, 3D printing does not yet have the same volume as traditional procedures.
The Belgian Health Care Knowledge Center (KCE) has published a report of recommendations for the “Responsible use of High-Risk Medical Devices” made using 3D printing.
The study says that because of the lower number of medical cases using 3D printing, currently a conclusion cannot be reached as to the question of whether 3D printing is a better option.
Specifically a press release accompanying the report says, “there is no convincing evidence that 3D printed implants […] are at least as safe and effective as the ‘classic’ products” (translation from original PR).
As part of a much larger 5-chapter document, the statement is simply a trigger to help encourage manufacturers, healthcare professionals and insurers to prepare for 3D printing as a medical eventuality, much in line with the FDA’s guidelines on the subject.
What is and isn’t included in Report 297
A thorough and systematic review, the report has been completed in consultation with around 40 external experts, referencing over 40 papers on the subject of 3D printing in medicine. It is also worth noting, that it exclusively refers to 3D printed implants, surgical guides and anatomical models. It does not cover 3D bioprinting, or 3D printed pharmaceuticals.
The making of the report
The majority of interviewed experts were representatives of Belgium’s Federal Agency for Medicines and Health Products (AFMPS) and the National Institute for Disability Health Insurance (RIZIV/INAMI).
Experts from academic hospital UZ Leuven, Materialise and international trade consultants beMedTech were also consulted, alongside individuals including Kevin Weatherwax, Administrative Program Director of Michigan State University’s College of Pharmacy, and Bernard Debbaut of Christelijke Mutualiteit, the largest health insurance fund in the country.
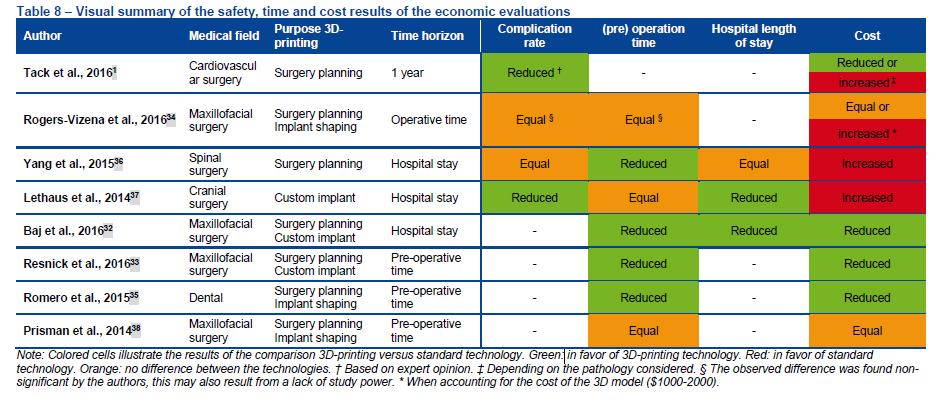
Too soon to tell
Despite such a comprehensive assessment of the safety and cost-effectiveness of 3D printed medical devices, a definite conclusion suffers due to lack of appropriate data.
The fact remains that there are still no standards specifically relating to 3D printing procedures, and the technology, when compared with traditional manufacturing, is still relatively new.
The KCE does however make a number of recommendations to policy makers, and particularly those in insurance, for suitable interim precautions.
First of all, “New 3D high-risk implants should initially only be used in certain specialized centers until there is more conscience about their safety and added value.”
Second, there should be more transparency between doctors and patients about the potential costs incurred through 3D printed treatment, as an insurance payout is not yet guaranteed.
And third, the KCE urges relevant government agencies to pool their resources, and create a more substantial database tracing the production of 3D printed implants, and results experienced by the patient.
The complete KCE Report 297 is a valuable addition to the literature around medical 3D printing, and at 164 pages worth a read.
Never miss out an another story – subscribe to the 3D Printing Industry newsletter, follow us on Twitter, and like us on Facebook.
Nominations for the second annual 3D Printing Industry Awards are now open. Make your selections for best medical application and more here.
Featured image shows a deconstruction of a cranioplasty procedure with 3D printed metal plate. Image via Renishaw.



