The 3D Printing Industry Awards 2022 shortlists are now available for voting. Who will win the 2022 3DPI Awards? Have your say by casting your vote now.
Dutch PEEK 3D printing start-up Bond3D and medical implant developer Invibio Biomaterial Solutions have come up with a means of additive manufacturing spinal cages that better facilitate patient recovery.
Together, the firms have managed to develop ‘next-generation’ spinal cages, which not only feature all the healing benefits of their predecessors, but can be configured with the porosity needed to stimulate new bone formation. Unveiled at Formnext, the firms’ cages are also said to have sufficient biocompatibility to gain FDA approval, and they’re currently going through the regulatory steps to gain this.
PEEK 3D printing at Bond3D
Operating out of its base in the city of Enschede, Bond3D specializes in production of functional components from high-performance polymers, certified for end-use in critical applications. The firm’s business revolves around a patented, pressure-controlled extrusion process, in which filament can be layered into prints with final-part densities of over 99%.
Due to its compatibility with materials like polyimides, fluoropolymers, liquid crystalline polymers, and polyaryletherketones (PAEKs), this technology allows Bond3D to address the medical, aerospace, energy, and automotive markets.
To help it further develop its unique approach to extrusion, Bond3D was awarded funding by Victrex in 2019, which was said to be valued at ‘millions of euros.’ Since then, the company has continued to build on its technology, unveiling a void-free PEEK 3D printing process, as well as announcing the build of eight production-ready systems, last year.
‘Fourth-generation’ spinal implants
Given that it’s sterilizable, highly biocompatible and features an impressive strength-to-weight ratio, PEEK has proven a popular material in the medical industry. In fact, Bond3D claims the polymer has already been used to create implants for over 15 million patients, whether they be in need of orthopedic, spine, trauma or cardiovascular treatment.
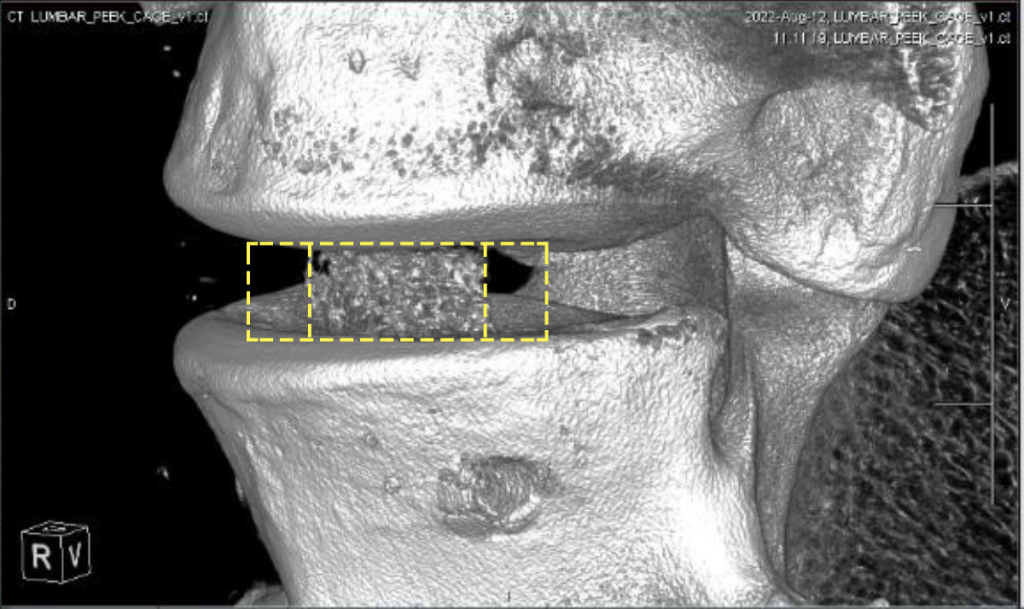
Where Bond3D has identified an opportunity in this field, is in the development of spinal interbody fusion devices, which allow for clearer artifact-free CT/MRI imaging. According to the company, producing implants with a modulus similar to natural bone, porous regions to encourage bone ingrowth and the rigidity to prevent cages caving in, also remains an ongoing challenge in the field.
To get around these imaging and stiffness-related issues, Bond3D has worked with Invibio Biomaterial to develop highly-porous spinal cage 3D printing capabilities. Bond3D says its technology allows for the creation of ‘fourth-gen’ implants that “combine the advantages of all previous generations,” and feature the enhanced biomechanical and biocompatibility properties needed to aid bone regrowth.
Currently, the companies are taking the final steps towards sending off an FDA submission for one of these devices with a US-based spinal implant developer. This submission is being supported by a master file, which Bond3D says will help any subsequent medical OEMs’ efforts to bring their designs to market in the future.

While 3D printing is still finding its feet in the wider medical market, the technology has already caught on in the field of custom spinal implant manufacturing. 4WEB Medical now markets a whole range of these devices, including its Stand-Alone Anterior Spine Truss System, designed to be used during spine disorder treatments.
As long ago as 2018, Johnson & Johnson acquired EIT, in a move that also saw the US multinational gain a strong foothold in the spinal implant 3D printing space. At the time, the deal was said to have strengthened the interbody implant portfolio of Johnson & Johnson’s orthopedics branch DePuy Synthes, which included titanium-integrated PEEK devices designed for minimally-invasive surgeries.
Check out our full Formnext news round-up for the latest from 3D printing’s largest trade show.
To stay up to date with the latest 3D printing news, don’t forget to subscribe to the 3D Printing Industry newsletter or follow us on Twitter or liking our page on Facebook.
While you’re here, why not subscribe to our Youtube channel? featuring discussion, debriefs, video shorts and webinar replays.
Are you looking for a job in the additive manufacturing industry? Visit 3D Printing Jobs for a selection of roles in the industry.
Featured image shows a CT image of a spinal cage printed by Bond3D. Image via Bond3D.



