Pioneering 3D printer manufacturer 3D Systems has announced that it has received 510(k) clearance from the FDA to 3D print maxillofacial surgical guides using its LaserForm Ti and DuraForm ProX PA materials.
According to the company, the new material choices will enable users to 3D print more innovative, lower profile surgical guide designs as part of its Virtual Surgical Planning (VSP) service. This will serve to give its customers, surgeons, greater access and visibility to the surgical site, reducing both operating times and the risks involved with maxillofacial surgery.
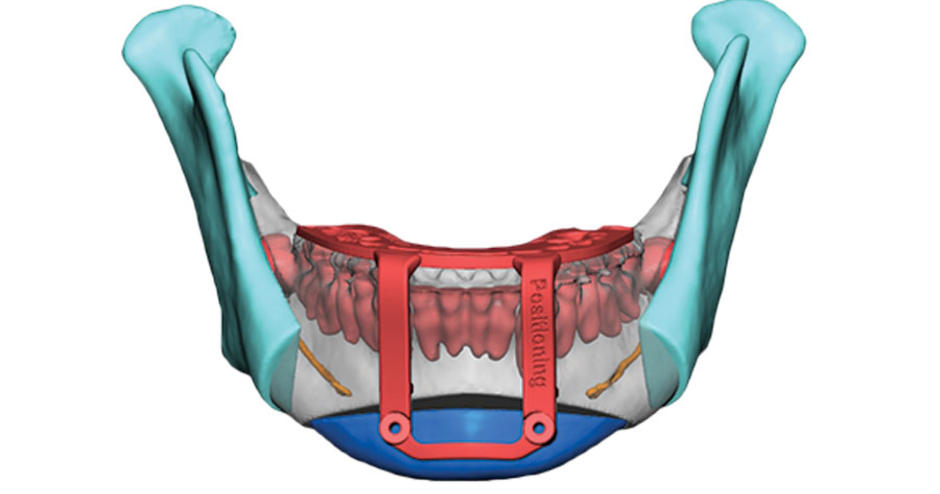
The Virtual Surgical Planning service
3D Systems’ VSP is a service-based approach to personalized surgery, drawing on the company’s expertise in medical imaging, surgical simulation, and additive manufacturing. Since 2018, it has been offered exclusively in collaboration with craniomaxillofacial product specialist Stryker’s CMF division.
Surgeons interested in the process present their desired surgical plans, along with any medical scans, in an online video meeting with a member of the 3D Systems biomedical engineering team. Together, they plan and simulate the surgical procedure to generate a ‘digital plan’. The VSP team can then 3D print patient-specific models, guides, and templates for the surgeon to take into the operating room. To date, the service has helped out in more than 120,000 patient cases around the globe.
Menno Ellis, Executive VP of Healthcare Solutions at 3D Systems, states: “Through close collaboration with surgeons and Stryker’s CMF division, we’ve uncovered opportunities to refine VSP guide designs that leverage additional capabilities in our materials portfolio. Our expert biomedical engineers are now able to design surgical guides tailored to the surgeon’s needs with enhanced properties that can help improve accuracy and facilitate procedures in ways not previously possible. Our powerful VSP System continues to transform surgery – enabling better patient outcomes.”
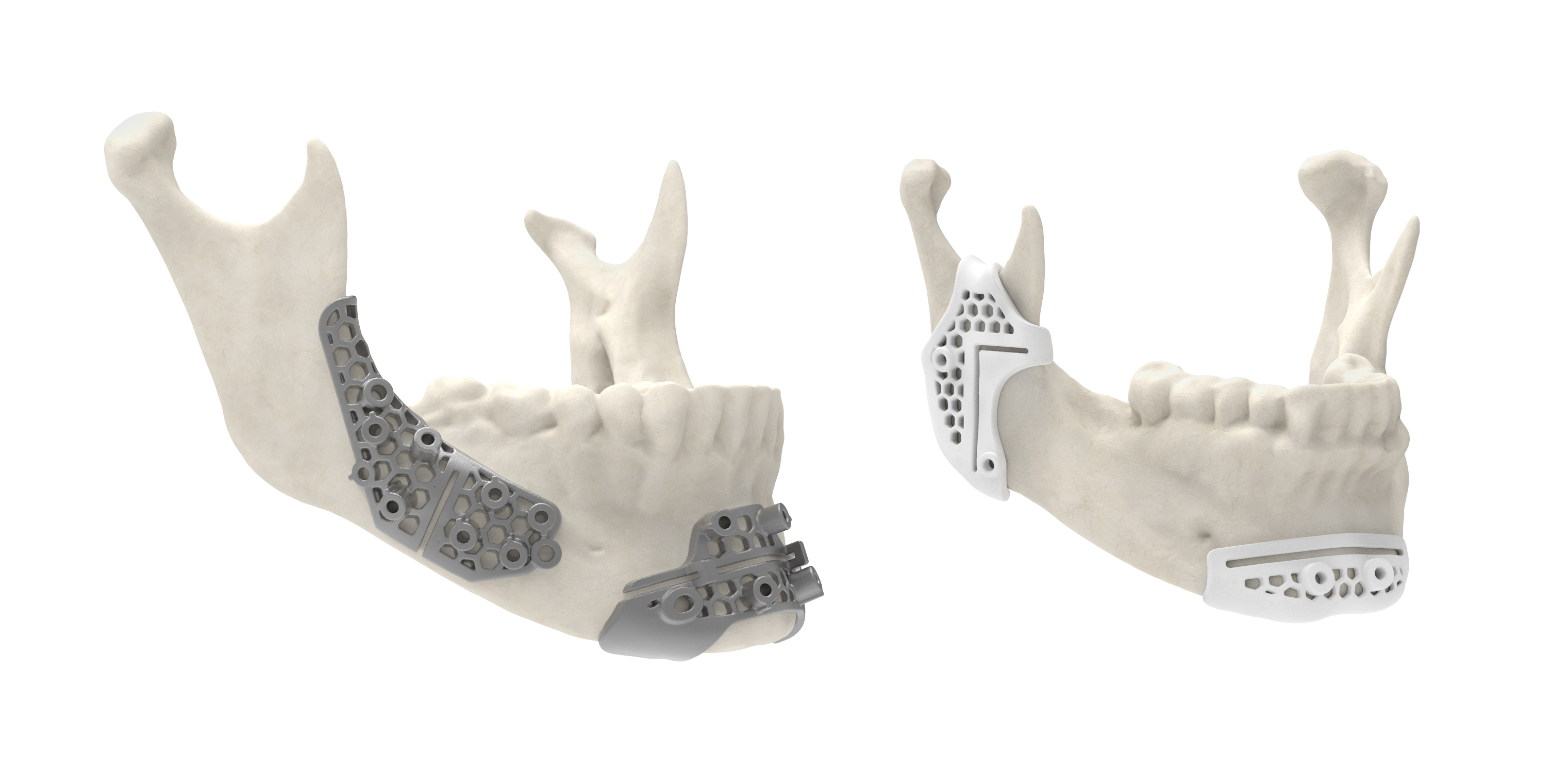
The LaserForm Ti and DuraForm ProX PA materials
The new material choices will allow the VSP service to print surgical guides with less overall material bulk, all while improving their strengths and rigidities. According to the company, mechanical tests have previously shown that cutting and marking guides 3D printed in LaserForm Ti can be up to 20x stronger than their conventionally manufactured titanium counterparts. The strength increase means they can be around 70% thinner, granting easier access to the critical surgery site.
Marking guides 3D printed in DuraForm ProX PA can also deliver toughnesses up to 88% higher than standard nylon ones, enabling them to withstand greater forces during the procedure. To top it off, both materials have been validated as safe for a wide range of sterilization methods. Surgeons are able to employ manual cleaning, automated cleaning, and steaming using a wrap or pouch to ensure the devices are patient-ready before entering the operating room.

With the development of standards and advancements in technological capabilities, the 3D printing of medical devices has accelerated in recent years. Just last month, ACS Custom, a specialist in customized hearing protection, announced that it was implementing 3D Systems’ Figure 4 system into its facility to upgrade its production of hearing aids. The company has stated that it intends to produce its small-scale in-ear devices using Figure 4 PRO-BLK resin.
Elsewhere, digital manufacturing service provider Fast Radius has previously partnered with Axial3D, a UK-based medical technology firm, to launch a ‘DICOM-to-print’ service for surgeons and hospitals across North America. The project will give clinicians access to improved surgical planning with micro-millimeter accurate, patient-specific 3D printed anatomical models.
Subscribe to the 3D Printing Industry newsletter for the latest news in additive manufacturing. You can also stay connected by following us on Twitter and liking us on Facebook.
Looking for a career in additive manufacturing? Visit 3D Printing Jobs for a selection of roles in the industry.
Featured image shows 3D printed titanium surgical guide. Image via 3D Systems.



