A Spanish-led research team has 3D printed a hydrogel capable of mimicking the behaviour of human lymph nodes, and accelerating the production of T-cells in cancer patients.
By combining a polyethylene glycol (PEG)-based polymer and the anticoagulant heparin, the team fabricated a structure which allowed T-cells to migrate and proliferate more effectively. Given that T-cells have the capacity to kill tumor cells, the research team’s new material could be used as a basis for a new form of cancer immunotherapy. The scientists have already filed a patent for their novel polymeric hydrogel with the European Patent Office, and they hope to bring their technology into hospitals in the near future.
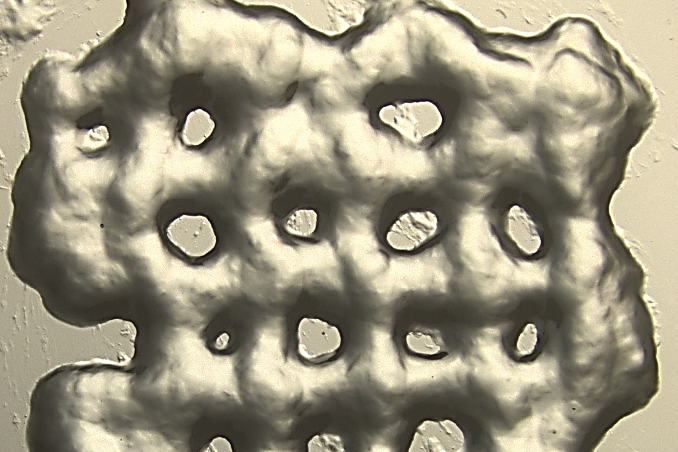
The need for a 3D printed T-cell solution
As opposed to chemotherapy and surgical treatments of cancer, immunotherapy harnesses the immune system of the patient to defend itself against the deadly disease. Cancer can appear within existing cells, and is capable of evading the immune system’s response, but T-cells can be programmed to identify and destroy such cells with varying levels of success.
T-cell based approaches face challenges though, as modified cells can be difficult to manufacture, manipulate, and control in large enough doses to fight cancer effectively. Additionally, treatment based on Tumor Infiltrating Lymphocytes (TILs), which actively seek out and destroy tumors, badly needs cell expansion systems to drive their wider adoption.
Previous research has primarily focused on the ex vivo proliferation of T-cells, but these approaches have failed to take the impact of human lymph nodes into consideration. Cytokine CCL21 for instance, a chemical present in the lymph node, interacts with naïve T-cells through the chemokine receptor CCR7, and enhances cell proliferation and migration.
3D printed Extracellular Matrices (ECMs) meanwhile, are often used to mimic genuine ECMs, and aim to create a nurturing environment that encourages cell expansion. In addition to providing more realistic conditions than standard petri dishes, fabricated ECMs can also be customized with different proteins and growth factors to enhance cell growth.
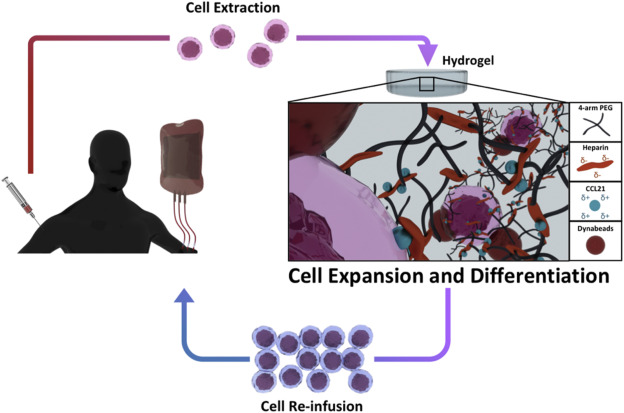
The Spanish research team’s novel approach
The researchers combined the benefits of CC21, a 3D printed PEG-based ECM and the blood thinner Heparin, to create an ECM that recreated the conditions found within the lymph node. If successful, the team’s hydrogel could actively increase CD4 T-cell proliferation, while fine tuning cell differentiation for Adoptive Cell Therapy (ACT).
In order to create their ECM hydrogel, the Spanish research team functionalized heparin with a maleimide chemical. The mixture was then left overnight, before being purified by dialysis, and combined with a PEG composite covered in Phosphate Buffered Saline (PBS). As a result of the amalgamation, covalent crosslinking took place, leading to gelation and ultimately, the creation of the team’s novel hydrogel.
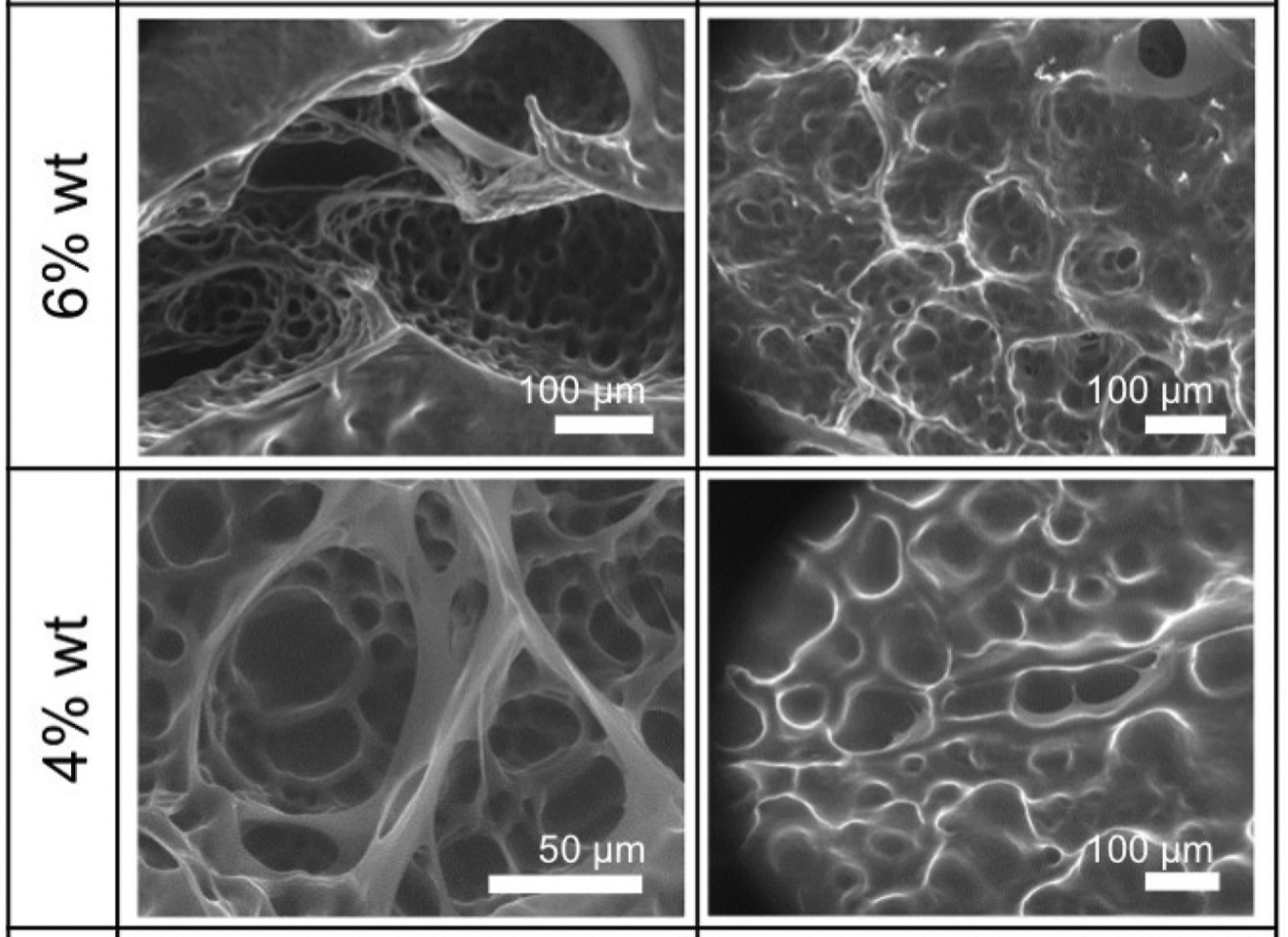
ECMs with different weight percentages of 6% wt, 4% wt and 3% wt were later fabricated, and subsequent rheology showed that each took 200-240 minutes to stabilize. The three different formulations were studied using Scanning Electron Microscopy (SEM) methods, and results revealed a correlation between low amounts of PEG, and a high level of porosity.
Further analysis showed that the median pore size observed was 55 μm, and that ECMs with this level of porosity or lower would be more conducive to cell expansion than thicker matrices. As a result, the 3% wt hydrogel was identified as the most hospitable to T-cell aggregation and proliferation. Different concentrations of 1 ng/ml, 20 ng/ml, and 100 ng/ml CC21 cytokine were then added to the ECM mix, to evaluate CD4 T-cell expansion.
A summary Propidium Iodide (PI) viability test showed that the T-cells replicated 25 percent more in the PEG-ECM, than they would in an ex situ suspended state. Cytokine-loaded hydrogels were also found to promote an increased proportion of effector cells, meaning that the makeup of T-cells could be modified in future to increase their toxicity to cancer cells.
The research team concluded that their approach had proved a valid alternative to ex vivo cell proliferation techniques. Adding CCL21 to the team’s hydrogel was also demonstrated through various evaluations to encourage cell growth. As a result, the researchers surmised that other chemical molecules, such as CCL19 and ICAM-1, could potentially lead to better clinical outcomes for patients with further research.
In future, the Spanish-led team believe that their novel PEG-based hydrogel could be widely-adopted within hospital settings. “The scaffold provides an improvement in cell proliferation and an influence on the phenotype, which could be further explored towards the fabrication of artificial lymph nodes,” stated the team in their paper.
“The CCL21-loaded 3D PEG-Hep hydrogels are also expected to be compatible with perfusion systems and novel bioreactors to work under Good Manufacturing Practice (GMP) conditions in large facilities,” they added.
3D printed Extracellular Matrices (ECMs)
3D bioprinted cell scaffolds have been leveraged by both pharmaceutical companies and university researchers in recent years, for a wide range of scientific innovations.
South Korean pharmaceutical company HK inno.N is working with 3D bioprinting firm T&R Biofab to test skin disorder drugs using the latter’s ECM bio gel matrices. If the evaluations are successful, the fabricated flesh could be used as an alternative to animal testing.
Researchers from the University of Minnesota have developed a novel ECM-based bio gel, allowing them to create a miniature 3D printed beating human heart. The team’s cell-laden biomaterial, produced using pluripotent stem cells, allowed them to fabricate a more detailed aortic replica.
Scientists from Oxford University and The Chinese University of Hong Kong devised a novel 3D bioprinting method, which allowed them to better understand how the human brain develops. By 3D printing human cortical cells into a soft, biocompatible ECM matrigel, the researchers were able to precisely pre-pattern them into both natural and unnatural designs.
The project was led by researchers from ICMAB and IBEC, with the collaboration of VHIO and UIC. The team’s findings are detailed in their paper titled “Examining metastatic behavior within 3D bioprinted vasculature for the validation of a 3D computational flow model,” which was published in the Advanced Functional Materials journal. The report was co-authored by Eduardo Pérez del Río, Fabião Santos, Xavier Rodriguez Rodriguez, Marc Martínez-Miguel, Ramon Roca-Pinilla, Anna Arís, Elena Garcia-Fruitós, Jaume Veciana, Joachim P. Spatz, Imma Ratera and Judith Guasch.
Nominations for the 2020 3D Printing Industry Awards are still open, let us know who is leading the industry now.
The fourth edition of the 3D Printing Industry Awards Trophy Design Competition is now underway. Enter your design for the chance to win a CraftBot Flow 3D printer.
To stay up to date with the latest 3D printing news, don’t forget to subscribe to the 3D Printing Industry newsletter or follow us on Twitter or liking our page on Facebook.
Are you looking for a job in the additive manufacturing industry? Visit 3D Printing Jobs for a selection of roles in the industry.
Featured image shows one of the Spanish team’s 3D printed PEG-based ECM scaffolds. Image via the Biomaterials journal.


