Professor Michael Kühl at the Department of Biology, University of Copenhagen (UCPH), and an international team of researchers from the Technical University of Dresden (TU Dresden), located in Germany, have developed a novel 3D bioprinting method for monitoring complex tissue structures.
Published in the latest edition of the materials science journal, Advanced Functional Materials, this method uses bioinks containing chemical sensor particles to enable noninvasive mapping of the metabolic activity of cells growing in bioprinted constructs.

Enabling online monitoring of cell activity
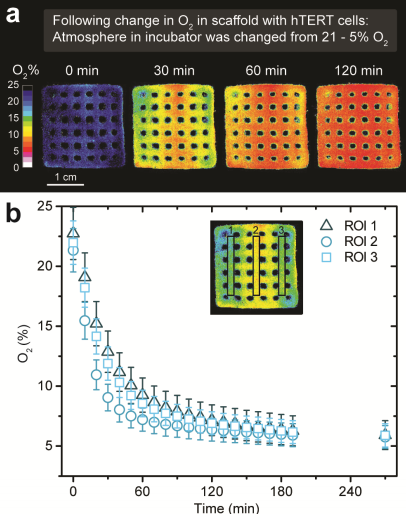
According to the study, monitoring cell growth in 3D bioprinted structures is important when evaluating its activity and reaction to chemicals. Bioprinting is combined with online imaging of oxygen by “functionalizing a hydrogel bioink via addition of luminescent optical sensor nanoparticles.” However, cell metabolism is mostly measured with media surrounding the constructs or by destructive sample analyses.
Versed in aquatic microbial ecology, Professor Kühl and a group of researchers, sought to address the scarcity of bioprinting measurement methods to facilitate rapid evaluation of cell activity in printed constructs. Professor Kühl stated:
“Living cells can be 3D printed in biocompatible gel materials (bioinks) and such 3D bioprinting is a rapidly developing field, e.g. in biomedical studies, where stem cells are cultivated in 3D printed constructs mimicking the complex structure of tissue and bones.”
“Such attempts lack online monitoring of the metabolic activity of cells growing in bioprinted constructs; currently, such measurements largely rely on destructive sampling. We have developed a patent-pending solution to this problem.”
Creating a novel functionalized bioink
Confronting this problem, the researchers developed a functionalized bioink with luminescent oxygen sensitive nanoparticles into the print matrix. The study explains that “blue light excites the nanoparticles, they emit red luminescent light in proportion to the local oxygen concentration – the more oxygen the less red luminescence.” Thus, with the distribution of red luminescence and oxygen, bioprinted living structures can be imaged with a camera system.
Eliminating the need for destructive sampling, this process allows for online monitoring of oxygen distribution and dynamics that can be mapped to the growth and distribution of cells in the 3D bioprinted construct. This method benefits regenerative cell research which uses bioprined 3D scaffolds as it conserves “structural complexity and metabolic interactions in mixed-species bioprints in external incubation conditions.” Professor Kühl explained:
“It is important that the addition of nanoparticles doesn’t change the mechanical properties of the bioink, e.g. to avoid cell stress and death during the printing process. The nanoparticles should not inhibit or interfere with the cells. Our method shows good biocompatibility and can be used with microalgae as well as sensitive human cell lines.”
According to Professor Kühl, 3D bioprinting with functionalized bioinks is a new powerful technology that can be applied in many other research fields than biomedicine, such as biophotonics. The team is now interested in exploring new collaborations and applications of their developments.
The research paper “Functionalized Bioink with Optical Sensor Nanoparticles for O2Imaging in 3D‐Bioprinted Constructs” is co-authored by Michael Kühl, Erik Trampe, Klaus Koren, Ashwini Rahul Akkineni, Christian Senwitz, Felix Krujatz, Anja Lode and Michael Gelinsky.
For more of the latest research papers on 3D printing subscribe to the 3D Printing Industry newsletter. Also, follow us on Twitter, and like us on Facebook.
Looking for new talent or seeking a career change? Search and post 3D Printing Jobs for opportunities and new talent across engineering, marketing, sales and more.
Featured image shows a 3D bioprinted structure and its imaging containing green algae (Chlamydomonas) in a hydrogel. Photo via Anja Lode/TU Dresden.

