One of the primary goals of The March Lab at Cornell University, New York, is to create synthetic intestines for use in regenerative medicine and animal-free drug testing. Made from stem cells, the artificial models are theoretically much closer to the real-thing than the alternatives, including computational simulation and single layer cell tests.
Now in the lab’s most recent study, published online October 2017, the team has applied 3D printing to its creation of a realistic intestine, using PolyJet technology to make a microscopic bioreactor.
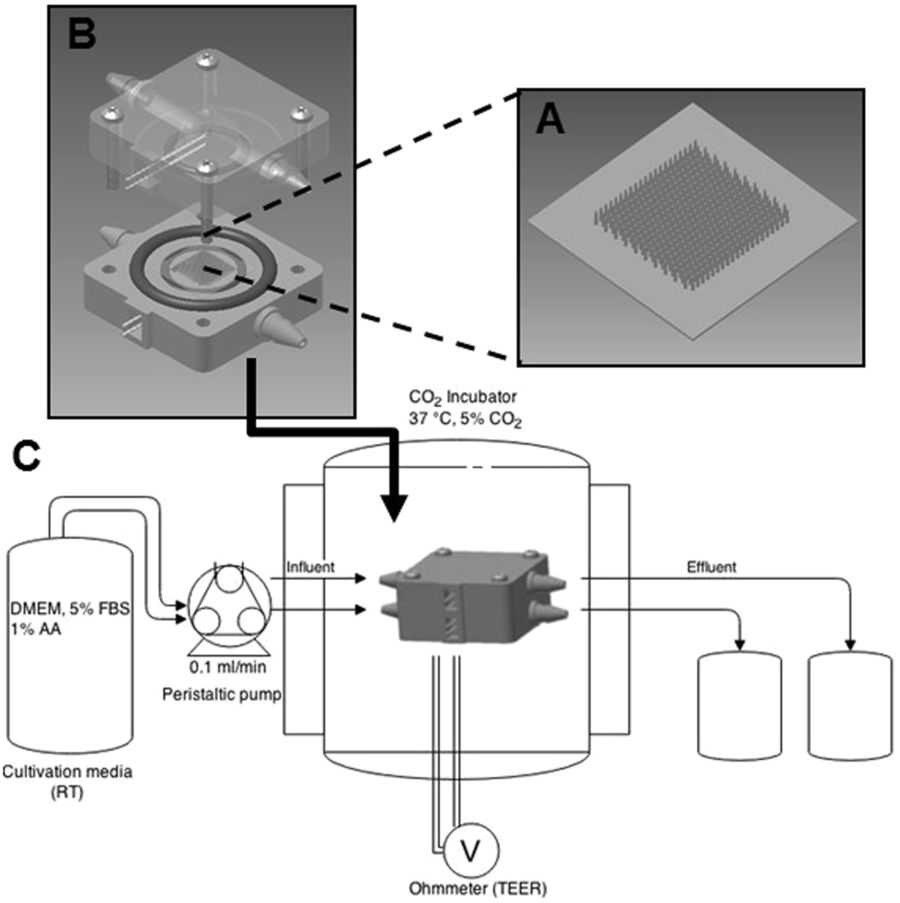
Going with the flow
The March Lab’s 3D printed bioreactor is responsible for inducing peristaltic fluid flow to cells found lining the intestine. The peristaltic fluid used is in turn responsible for the contraction and relaxation of muscles, making culture conditions in the Cornell bioreactor more lifelike than those without flow.

The device has been fabricated on a Stratasys Objet30 Pro 3D printer using VeroClear-RGD810 resin, and a CAD file modeled using Autodesk software.
Stamped cell scaffolds
Cells lining the intestine are called lumen. The synthetic lumen in the March Lab’s is supported by a cell scaffold. The scaffold is made by March Lab researchers using a micromolding process, that effectively stamps a negative of the undulating peaks and troughs of a lumen into acrylic, then casts the shape in a suitable material.
Schematic animation demonstrating Cornell’s intestinal scaffold micromolding process. Clip by Michael Campolongo
Once fabricated, the scaffold is laced with intestinal cells and placed in the bioreactor for culture. In this experiment, March Lab researchers compare the results of culture inside the 3D printed bioreactor to cells grown in static conditions. As evident from the image below, cells grown inside the reactor are more defined and uniform to those grown without induced peristaltic fluid.

Functionality of the cells was also assessed for an ability to absorb glucose. Results showed that cells cultured in the bioreactor had “significantly more uptake than in static systems” and “Importantly, levels reached are consistent with estimates for physiological conditions.”
Trusting the gut
According to the study’s conclusion, “We anticipate that such small intestinal bioreactors will be useful for future studies of intestinal function, including high throughput drug absorption profiling and studies of bacteria-host interactions.”
“The more we understand about how our guts work, the more we can develop therapies and gain insights into the evolution of human biology.” Professor John March, Cornell University.
Other organs to have been modeled in a similar way include the kidney, the heart and the lungs. By connecting such devices, other researchers are working towards developing an entire micro model of the human body, giving a more accurate picture of how drugs react in the body. The research could be revolutionary for discovering new treatments, especially for cancerous diseases.
Speaking to the Cornell Chronicle, lab lead Professor John March explains,”The intestine is home to most of our immune system and it is also where all of our nutrients are absorbed, so to function as a human being or any multicellular animal, our gut is the centerpiece of our interactions with the outside world.”
“The more we understand about how our guts work, the more we can develop therapies and gain insights into the evolution of human biology.”
The research discussed in this article is co-authored by Cait M. Costello, Mikkel B. Phillipsen, Leonard M. Hartmanis, Marek A. Kwasnica, Victor Chen, David Hackam, Matthew W. Chang, William E. Bentley and John C. March. A full study of the findings is published online in the journal Scientific Reports.
For more 3D printing research delivered daily, subscribe, like and follow. Register for 3D printing jobs here.
Featured image shows SEM image of intestinal lumen cells grown in the 3D printed bioreactor (A) vs. cells grown in static conditions (B). Image via Scientific Reports


