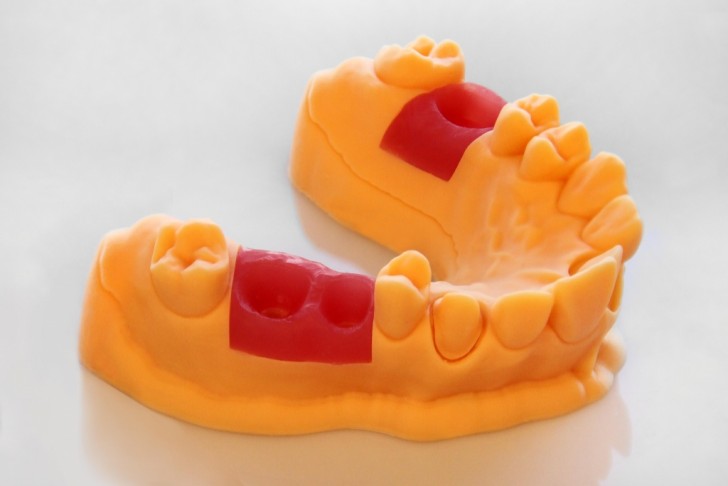
Not many 3D printing companies have the expansive reach of Germany-based company EnvisionTEC, who have helped to innovate a number of premium 3D printing materials, including (but certainly not limited to) 3D printed electronics, bioprinted metals, and even wax. Now, EnvisionTEC is taking aim at the dental industry with their newest 3D print material, which has just already been approved for use by the FDA. Their dental-driven materials include wax-filled polymers (Press-E-Cast), which can be used for detailed coping and crown production, as well as the FDA approved E-Guard (for bite guards) and also the E-Gum material, which can be used when producing crowns and bridges over a dental model with removable dies.
“We’re thrilled to be able to announce the latest addition to our innovative line of premium 3D print materials at the largest dental laboratory event in America,” said EnvisionTEC CEO Al Siblani. “Our new material can be printed on our entire Perfactory® and Vida 3D Printer lines and is the perfect material for high throughput dental applications, providing on-site turnkey solutions for dental labs and offices.”
These newly developed materials are already compatible with a wide-range of EnvisionTEC’s 3D printing solutions. Their Perfactory Vida 3D Printer Series, for example, can be utilized to 3D print numerous dental and orthodontic models, bite splints, retainers, and surgical guides. Their newly released Vida Crown & Bridge printer, on the other hand, can be used to create partial dentures, as well as (which I’m sure you can infer from the name) crown and bridge work too. A number of other EnvisionTEC 3D printing materials make-up their well-rounded digital dentistry solutions:
-
The EnvisionTEC Vida 3D Printer E-Partial – designed to enable the creation of partial denture frameworks for casting
- Clear-Guide – optimized for the creation of highly accurate, transparent drill guides for oral surgery
- Ortho Tough – a tough resin for use in high temperature situations such as the creation of orthodontic aligners by vacuum thermoforming
- E-Guard – FDA-approved, biocompatible and transparent for the production of accurate bite splints and night guards
- E-Appliance – ideal for printing orthodontic models. It is a specially designed nano-filled resin that works with both polyvinyl alcohol-based separators and a tin foil substitute for a “salt and pepper” technique
EnvisionTEC will be showcasing their new E-Guard and E-Gum materials at the LMT LAB DAY in Chicago, Illinois, which is taking place this weekend at the Hyatt Regency. The company booth will include all of their digital dentistry solutions at the expo, and will be located at booth A-43/B-42 throughout the weekend. You can get a preview of the new flexible material at LMT LAB DAY. Personally, I’m astounded by the amount of industries that EnvisionTEC has innovated upon with their 3D printing technology, and now, they are bound to cause even brighter smiles with their newest dental-driven 3D printing materials.
Correction: We originally reported that the E-Gum material was FDA approved, but that is not the case. It is the E-Guard and E-Dent materials that are FDA approved for use in the mouth. It was explained by EnvisionTEC’s Senior Communications Specialist Jenna Franklin that the E-Gum is meant to simulate the way the gum will affect the crown or bridge’s fit. It is placed over the model in the location where the die will be inserted. But can’t go into the actual mouth. The necessary corrections have been made.